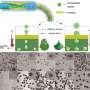
Rapid and efficient oil-water separation achieved by newly-developed particles

Chinese researchers have developed new magnetic Janus particles for oil-water separation. These particles—hydrophilic/oleophilic magnetic Janus particles—can separate micro-scaled oil droplets from water rapidly and efficiently.
Oil pollution has become a global challenge. In practice, oils are usually classified as floating oils, dispersed oils, surfactant-stabilized oil droplets and surfactant-free micro-scaled tiny oil droplets. In recent years, many efforts through control of the surface wettability towards achieving the effective separation of floating oils, dispersed oils and even surfactant-stabilized oil droplets have been made. However, the separation of micro-sized oil droplets from water has often been overlooked.
Micro-scaled oil droplets with diameters less than 20 μm are very stable in water. Many challenges remain when using traditional methods and superwettable materials to separate these micro-scaled oil droplets from water.
Recently, researchers from the Technical Institute of Physics and Chemistry of the Chinese Academy of Sciences (TIPCCAS) have developed an emulsion interfacial polymerization approach to synthesize anisotropic Janus particles with controllable topological and chemical anisotropy.
This method can also be expanded to large-area fabrication of two-dimensional Janus film actuators.

On the basis of these studies, the researchers recently synthesized hydrophilic/oleophilic magnetic Janus particles by emulsion interfacial polymerization and the interfacial assembly of Fe3O4 nanoparticles, realizing the rapid and efficient separation of micro-scaled tiny oil droplets from water.
When adding the hydrophilic/oleophilic magnetic Janus particles to the oil-in-water emulsion and subsequently shaking for one minute, the originally stable emulsion rapidly formed layers.
After manipulation with a magnetic field, the upper oil phase could be rapidly attracted toward the magnet. The whole process was accomplished in two minutes. The separation achieved a high efficiency (>99%) and is applicable to various oils and various oil contents.
Researchers also found that the oleophilic surface of Janus particles was beneficial for capturing a large number of tiny oil droplets to make them coalesce. Finally, like surfactant molecules, these Janus particles could adsorb at the interface of large oil droplets to stabilize them.
These Janus particles can be self-assembled on the interface of the larger oil droplets with their oleophilic concave surface toward the oil phase and their hydrophilic convex surface toward the water phase.
These particles provide a good candidate for application in the clean-up of industrial wastewater and water purification.
More information: Yongyang Song et al, Hydrophilic/Oleophilic Magnetic Janus Particles for the Rapid and Efficient Oil-Water Separation, Advanced Functional Materials (2018). DOI: 10.1002/adfm.201802493
Journal information: Advanced Functional Materials
Provided by Chinese Academy of Sciences