CryoEM study captures opioid signaling in the act
Opioid drugs like morphine and fentanyl are a mainstay of modern pain medicine. But they also cause constipation, are highly addictive, and can lead to fatal respiratory failure if taken at too high a dose. Scientists have long sought to develop new opioid drugs that can drive away pain without these dangerous side effects, but holes in our understanding of exactly how opioids exert their various effects at a biological level has so far kept this dream at bay.
Opioid painkillers work by binding to a receptor protein present on nerve cells called the µ-opioid receptor, which evolved to respond to the body's natural painkillers (such as the endorphins produced by exercise) by tuning down pain and creating a sense of euphoria. Opioid drugs from opium to morphine to heroin hijack this signaling system by binding to the same receptor molecule. But details of how activating these receptors triggers the drugs' positive and negative effects have remained hazy.
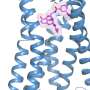
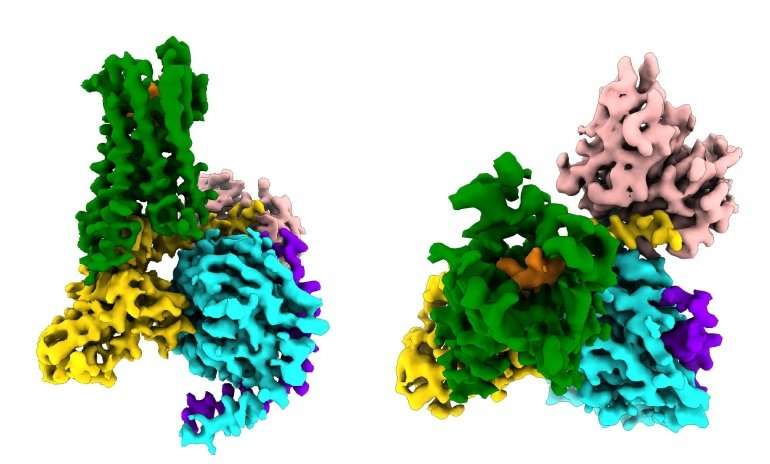
Now, in a study published on June 13, 2018 in Nature, scientists at UC San Francisco and Stanford University have used ultra-high-resolution cryo–electron microscopy (cryoEM) to capture the most detailed portrait ever of an opioid drug triggering the biochemical signaling cascade that gives it its power – both for good and for ill.
"We've essentially captured this signaling event in the act," said study co-senior author Aashish Manglik, MD, Ph.D., an assistant professor of pharmaceutical chemistry in UCSF's School of Pharmacy who conducted the new study as a graduate student and Distinguished Fellow at Stanford. "These new atomic-level images will hopefully enable us to rationally design compounds that target different aspects of opioid signaling in the brain, with hopes of identifying new, safer painkillers."
The µ-opioid receptor is part of a large family of hundreds of signaling proteins called G-protein coupled receptors (GPCRs) that are involved in everything from vision and hearing to the immune system's response to invasive pathogens, and are the targets of more than 30 percent of modern drugs. Most GPCRs share the same basic mechanisms: When the right signaling molecule (for example, an opioid) binds to a GPCR on the outside of the cell, the protein stimulates a chain reaction of biochemical signals within the cell by activating a messenger molecule called a G protein (hence the name GPCR).
Experiments that revealed how a different type of GPCR binds to the "stimulatory" G protein led to a Nobel Prize for Stanford's Brian Kobilka, MD, one of the senior authors of the new study, but researchers have known for decades that GPCRs can also bind to as many as a dozen other signaling molecules within the cell. For example, µ-opioid receptors typically only activate so-called "inhibitory" G-proteins, which have the opposite effect of the stimulatory G protein cascade. However, scientists aren't sure what causes some GPCRs' affinity for particular partner proteins within the cell, or exactly what the consequences are.
Researchers hope that by understanding these different pathways of GPCR signaling, they may be able to develop drugs with highly specific effects, such as suppressing pain without causing addiction. But until now, researchers had little idea how a given GPCR selectively interacts with only a subset of signaling partners within the cell.
The new study, published June 13, 2018 in Nature, captured for the first time how the µ-opioid receptor binds to its inhibitory G protein partner. Among other findings, the study showed that the receptor's selectivity appears to be due to the small size of the binding pocket for the G protein on the inside of the cell, whereas the stimulatory G protein requires a larger binding site.
Manglik previously collaborated with the computational drug discovery lab of Brian Shoichet, Ph.D., a professor of pharmaceutical chemistry in UCSF's School of Pharmacy, to identify a molecule called PZM21 that allows the µ-opioid receptor to only engage the inhibitory G protein but not another signaling molecule called beta-arrestin, and showed that this selective drug provided pain relief with reduced side effects in mice. His lab is now building on the new, high resolution portrait of opioid receptor – G protein complex to develop new, even more selective compounds.
More information: Antoine Koehl et al. Structure of the µ-opioid receptor–Gi protein complex, Nature (2018). DOI: 10.1038/s41586-018-0219-7
Journal information: Nature
Provided by University of California, San Francisco