Chemical complex can enhance excretion of radioactive strontium from the body
90Sr (t1/2 = 29.1 y) is one of the most important nuclear fission elements. After the nuclear power plant disasters of Chernobyl and Fukushima Daiichi, 90Sr was released into the atmosphere and the ocean. 90Sr is harmful to humans because it has a long half-life, and its daughter radionuclide, 90Y (t1/2 = 64.1 h), emits high-energy beta particles. It has been reported that internal exposure to 90Sr could be associated with the development of leukemia and osteosarcoma. Therefore, compounds inhibiting the absorption of radiostrontium from the gastrointestinal tract into the bloodstream and enhancing its elimination after intake can decrease the absorbed radiation dose of people exposed to radiostrontium. Indeed, basic research has shown that alginate can promote the excretion of 90Sr.
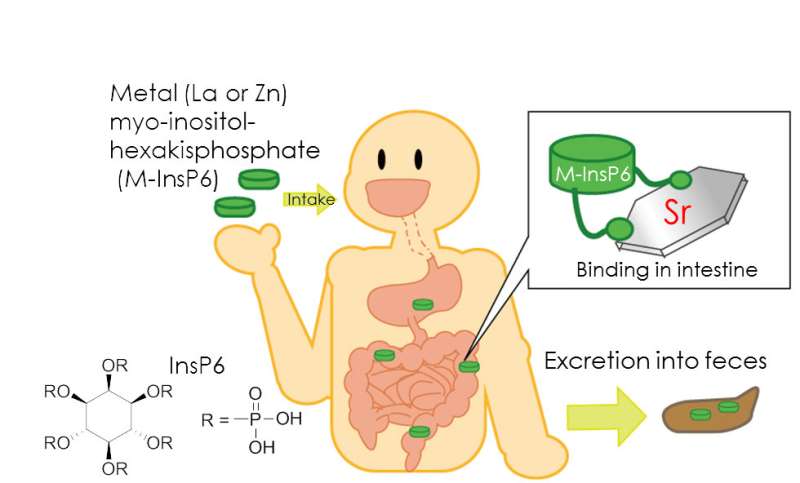
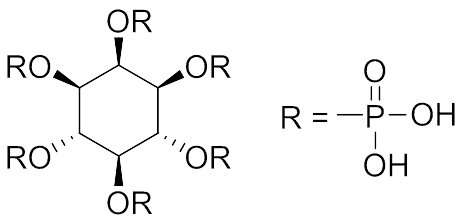
Myo-inositol-hexakisphosphate (phytic acid: InsP6, Figure 1) is a natural compound that abounds in plants, especially in whole grains, cereals, legumes, seeds and nuts. Due to its structure, InsP6 exhibits high chelation potential with many kinds of metal cations. Recently, we prepared complexes of InsP6 with zinc or lanthanum ions (Zn-InsP6 and La-InsP6, respectively) and evaluated them as radiocesium decorporation agents, because both complexes are insoluble in water and are spacious enough to accommodate additional metal cations coordinated by chelation. In fact, InsP6 is soluble in water, but chelation with zinc or lanthanum ions decreases the solubility in water, and thereby reduces the absorption in the intestinal tract.
In this study, we hypothesized that Zn-InsP6 and La-InsP6 can work as 90Sr decorporation agents and evaluated their potential in vitro and in vivo. Experiments using normal mice were performed to evaluate the influence of Zn-InsP6 on the biodistribution of radiostrontium. In these experiments, we replaced 90Sr with 85Sr (T1/2 = 64.8 d) because 85Sr emits gamma rays, which are easy to measure.
Adsorption capacity of Zn-InsP6 and La-InsP6 for strontium was evaluated by the Langmuir model. The amount of strontium bound to Zn-InsP6 and La-InsP6 increased linearly at low strontium concentrations, and plateaued at high strontium concentrations, indicating saturation of the adsorption sites (Figure 2). These data meet the requirements of the Langmuir adsorption model, indicating the correctness of the assumption that the adsorbate acts as a uniform surface with finite identical binding sites characterized by monolayer adsorption of the adsorbate. The maximum sorption capacity of Zn-InsP6 and La-InsP6 was estimated as 133.7 and 6.4 mg Sr/g, respectively.

To evaluate whether Zn-InsP6 can enhance the elimination of 85Sr from the body in vivo, 85SrCl2 was orally administrated to mice just after oral administration of Zn-InsP6 suspension. As the results, pretreatment with Zn-InsP6 significantly decreased the accumulation of radioactivity in the bone and the blood after oral administration of 85SrCl2. These results indicate that Zn-InsP6 inhibited the absorption of 85Sr2+ from the intestine into the bloodstream and enhanced the excretion of 85Sr2+ in the feces.
Extrapolating from the results of the biodistribution experiments, the estimated effective dose of 90Sr radiation in a human treated with Zn-InsP6 is approximately half of that of the control group. Therefore, Zn-InsP6 presents as a promising 90Sr decorporation agent.
We suppose that the Zn-InsP6 complex could improve the biodistribution of some radiopharmaceuticals and decrease the unnecessary radiation dose of patients, and it is currently under investigation.
More information: Kazuma Ogawa et al, Complexes of myo-inositol-hexakisphosphate (InsP6) with zinc or lanthanum to enhance excretion of radioactive strontium from the body, PLOS ONE (2018). DOI: 10.1371/journal.pone.0195067
Journal information: PLoS ONE
Provided by Kanazawa University