Zinc is able to modulate Escherichia coli bacteria's virulence, study finds
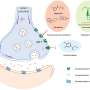
zinc can modulate the virulence of Escherichia coli bacteria, a pathogen that causes urinary infections in humans, according to a new study published in the journal Scientific Reports by biologist Carlos Balsalobre, from the University of Barcelona (UB). The new study reveals for the first time that zinc can regulate the expression of bacterial virulence factors, in particular, alfa-hemolysin, an exotoxin produced by some Escherichia coli pathogenic strains.
About 150 million cases of urine infection are diagnosed each year worldwide, most of them result from bacteria. This pathology, which is usually recurrent, affects more women than men and can cause health complications if they are not treated in time. Between 70 percent and 80 percent of the infections result from pathogenic strains of E. coli, a bacterial species with a wide range of virulence factors, including alfa-hemolysin, one of the most studied bacterial toxins, which has cytotoxic effects on cells). In particular, alfa-hemolysin produces cytotoxic effects on the bladder epithelial cells during the infection process.
Zinc is an essential metal for cell physiology. Cellular respiration, DNA and RNA replication, intracellular signaling and protein synthesis all depend on zinc. However, zinc is toxic in high concentrations, which is why the regulation of intracell zinc levels (homeostasis) is a precisely controlled machinery. Lecturer Carlos Balsalobre from the Department of Genetics, Microbiology and Statistics of the UB says, "The concentration levels of zinc in the intestine, urine and in intracellular levels can change in a wide range. Regarding the intestine, which has high concentrations of zinc, the alfa-hemolysin stops its expression, and pathogenic E. coli does not usually generate infections. However, if the metal concentration lowers, for instance, in the urinary tract, toxin expression increases. Also, during the infection process, the bacteria gets into the cells, where zinc concentrations can be even lower."
Promoting in vitro bacterial growth with no zinc has been one of the most complex methodological challenges in this new study. Therefore, it was necessary to remove the zinc with chelating elements and to add basic elements (except for zinc). Moreover, the collaboration with the team led by professor Thomas O'Halloran from Northwestern University (United States) worked with the purity of the zinc uptake regulator (ZUR) protein.
Zinc ads the ZUR transcription factor and favours its binding with DNA (in particular, in the gene-promoting area it regulates). Elsa Velasco, first author of the study, notes that "as a result of this binding, the transcription stops, and so does the gene expression. Therefore, when there is zinc, the ZUR-regulated genes are silenced. However, with a lack of zinc, the ZUR factor cannot join the DNA, and the regulated genes by this transcription factor do show expression."
According to the new study, "some genes which are codified by hemolysin have a binding site for the ZUR regulator in the promoting region," says Velasco. "Therefore, the gene expression of this toxin is affected by the zinc levels." Between 40 percent and 50 percent of E.coli pathogenic strains are able to express alfa-hemolysin. However, not all genes that are related to hemolysin production are zinc-responsive. So far, two types of genes taking part in the alfa-hemolysin synthesis in pathogenic E.coli have been identified. The new study, now published, shows for the first time that these systems are regulated in different ways.
Many bacteria are resistant to known drugs. Some bacterial infections, which were previously overcome with pharmacological treatments, no longer respond to antibiotics. Today, bacterial resistance is one of the main threats to global health, and a problem for patients suffering from recurrent urine infections. Knowing in greater detail about regulation mechanisms in the virulence factors of the pathogenic E.coli will enable researchers to discover how these bacteria act during the infection process—that is, which elements and strategies it uses to invade biological tissues. The new study, led by the UB, provides tools to work on new therapeutic treatments specifically designed to help treat some of the key phases of the bacterial infectious process.
More information: Elsa Velasco et al. A new role for Zinc limitation in bacterial pathogenicity: modulation of α-hemolysin from uropathogenic Escherichia coli, Scientific Reports (2018). DOI: 10.1038/s41598-018-24964-1
Journal information: Scientific Reports
Provided by University of Barcelona