A new tailor-made approach to biofuels

EU researchers designed and tested cell structures, cellulosomes, that help breakdown abundant biomass waste to produce value-added chemicals, such as advanced biofuels.
Key to producing advanced biofuels from lignocellulosic biomass is the efficient conversion of the cellulose contained within cell walls into fermentable sugars - a major bottleneck for large-scale production. Efficient breakdown of biomass into sugars, called saccharification, can reduce the cost of the process and lower also the amount of feedstock required for production of biofuels with milder or shorter pre-treatment time.
Some microbes have natural structures called cellulosomes that contain cellulases, enzymes that break down cellulose efficiently into sugars. The EU-funded CELLULOSOMEPLUS project developed designer cellulosomes (DCs) to achieve high yields of fermentable sugars from the organic fraction of municipal solid waste (OFMSW) to create advanced biofuels at a low process cost.
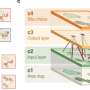
The consortium produced the basic components of natural cellulosomes as well as other lignocellulosic enzymes and, after their assembly into DCs, characterised the hydrolysis of the OFMSW substrate. They also studied the cellulosome's physicochemical, atomic and supramolecular structure, and the interactions of its various components. "Cellulosome architecture consists of a non-catalytic 'scaffoldin' subunit that carries 'cohesins' that are complementary recognition modules of another type of domain termed 'dockerins', which are carried by the enzymes and allow their integration into the complex," explains project coordinator Dr Mariano Carrión-Vázquez.
New assays and models developed
To characterise the cellulosomal and non-cellulosomal components to be used in the DCs, the team developed a standardised enzymatic assay. Another assay for determining enzymatic activity in industrial substrate showed that cellulosomes hydrolyse the lignocellulosic biomass in a more efficient manner than a single purified enzyme or a mixture of enzymes. "These studies provided a deeper understanding of the architecture, nanomechanics and catalytic properties of cellulosomes, and the logic behind their construction," says Dr Carrión-Vázquez.
Multi-scale modelling from atomic to supramolecular levels was used to investigate the structure and self-assembly of the cellulosome. According to Dr Carrión-Vázquez: "Models provided new information on the dynamic properties of catalysts together with mechanical stabilities, resulting in new ideas for experiments and in a synergistic feedback loop for producing optimised DCs."
The knowledge gained about natural cellulosomes and the subsequent screening of their components provided researchers with a platform to test the construction of the final DCs. These carried both cellulosomal and non-cellulosomal components and were validated at the laboratory and pre-industrial scale to ensure the optimum breakdown of the OFMSW industrial substrate.
Benefits for European biotechnology
Project partners analysed the cellulosomes of nine bacterial species and created a database of cellulosomal components. The thermal stability of key cellulosomal enzymes was increased by random and semi-rational mutagenesis, achieving better hydrolysis yields. Scientists also identified the 3D structures of three glycoside hydrolases and used the data to develop computational models. Different approaches to characterise interactions between cellulosomal components by single-molecule force spectroscopy based on atomic force microscopy methods were also developed, reducing sample preparation time and increasing data quality and comparability. Also, the mechanostability of scaffoldin cohesin modules was found to be a relevant new industrial parameter for the enzymatic activity of the cellulosome.
The consortium built several models of multi-domain enzymes and DCs presenting a detailed picture of cellulosomal assemblies. This included relevant data concerning the thermostability and substrate specificity of key cellulosomal elements.
The team developed a DC formed by ten components, combining cellulase/xylanase activities with three accessory enzymes. Furthermore, the complexation of enzymes into optimised DCs increased the hydrolysis on model substrates and pretreated lignocellulosic biomass. "The use of self-assembled DCs as nanocatalysts will benefit European biotechnology industries working in the transport-related sector, as well as chemical industries engaged in processing urban waste and residues from the agro-food, paper, and forestry sectors," observes Dr Carrión-Vázquez.
Tailoring DCs for the several biomass lignocellulosic residues should help improve process efficiency, thus lowering production costs. "This new technology will also help to reduce Europe's reliance on oil, strengthen SMEs within the EU, stimulate job creation and reduce the environmental impacts of the advanced biofuel sector."
Provided by CORDIS