Nanowires could make lithium ion batteries safer
From cell phones and laptops to electric vehicles, lithium-ion batteries are the power source that fuels everyday life. But in recent years, they have also drawn attention for catching fire. In an effort to develop a safer battery, scientists report in the ACS journal Nano Letters that the addition of nanowires can not only enhance the battery's fire-resistant capabilities, but also its other properties.
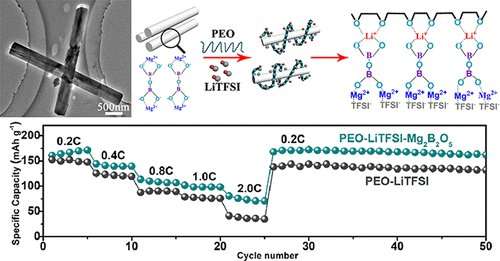
In lithium-ion batteries (LIBs), lithium ions move back and forth between electrodes through an electrolyte. Traditional LIBs have a liquid electrolyte made of salts and organic solvents, but it evaporates easily and can be a fire hazard. So, researchers have turned their attention to solid-state electrolytes as potential alternatives. Several options have been proposed for solid-state electrolytes, but most are not stable or cannot meet large-scale demands. Polymer electrolytes have shown potential because they are stable, inexpensive and flexible; but they have poor conductivity and mechanical properties. So, scientists have been adding an array of compounds to enhance the electrolyte. Xinyong Tao and colleagues previously made magnesium borate (Mg2B2O5) nanowires, which had good mechanical properties and conductivity. They wanted to see whether these properties would also be imparted to batteries when these nanowires were added to a solid-state polymer electrolyte.
The team mixed the solid-state electrolyte with 5, 10, 15 and 20 weight percent of the Mg2B2O5 nanowires. They observed that the nanowires increased the conductivity of the electrolytes and allowed them to sustain more stress compared to the electrolyte without nanowires. The increase in conductivity was due to an increase in the number of ions moving through the electrolyte at a faster rate. The group also tested the flammability of the electrolyte and found that it barely burned. When the nanowire-enhanced electrolyte was paired with a cathode and anode like it would be in a battery, the set-up had a better rate performance and higher cyclic capacity than batteries without added nanowires.
More information: Ouwei Sheng et al. Mg2B2O5 Nanowire Enabled Multifunctional Solid-State Electrolytes with High Ionic Conductivity, Excellent Mechanical Properties, and Flame-Retardant Performance, Nano Letters (2018). DOI: 10.1021/acs.nanolett.8b00659
Abstract
High ionic conductivity, satisfactory mechanical properties, and wide electrochemical windows are crucial factors for composite electrolytes employed in solid-state lithium-ion batteries (SSLIBs). Based on these considerations, we fabricate Mg2B2O5 nanowire enabled poly(ethylene oxide) (PEO)-based solid-state electrolytes (SSEs). Notably, these SSEs have enhanced ionic conductivity and a large electrochemical window. The elevated ionic conductivity is attributed to the improved motion of PEO chains and the increased Li migrating pathway on the interface between Mg2B2O5 and PEO-LiTFSI. Moreover, the interaction between Mg2B2O5 and −SO2− in TFSI– anions could also benefit the improvement of conductivity. In addition, the SSEs containing Mg2B2O5 nanowires exhibit improved the mechanical properties and flame-retardant performance, which are all superior to the pristine PEO-LiTFSI electrolyte. When these multifunctional SSEs are paired with LiFePO4 cathodes and lithium metal anodes, the SSLIBs show better rate performance and higher cyclic capacity of 150, 106, and 50 mAh g–1 under 0.2 C at 50, 40, and 30 °C. This strategy of employing Mg2B2O5 nanowires provides the design guidelines of assembling multifunctional SSLIBs with high ionic conductivity, excellent mechanical properties, and flame-retardant performance at the same time.
Journal information: Nano Letters
Provided by American Chemical Society





















