February 20, 2018 report
Using organoids to understand how the brain wrinkles
A team of researchers working at the Weizmann Institute of Science has found that organoids can be used to better understand how the human brain wrinkles as it develops. In their paper published in the journal Nature Physics, the team describes how they used a modified form of organoid development to study the development of brain wrinkles. Larry Taber with Washington University offers a News & Views piece on the work done by the team in the same journal issue.
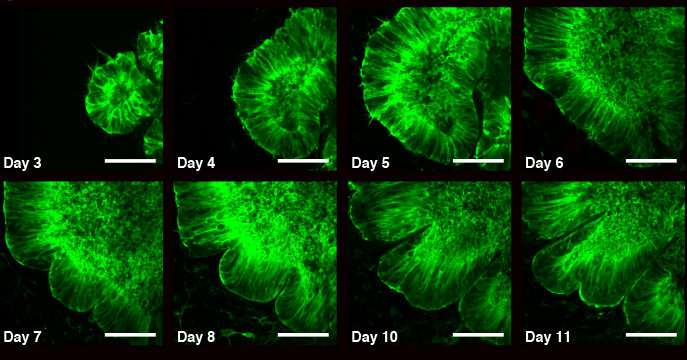
An organoid is an artificially grown mass of cells meant to replicate human or other animal organs. They are typically much smaller than the organs they are meant to mimic, but allow researchers a unique means of studying how organs develop. In this new effort, the researchers sought to better understand the process by which the human brain develops wrinkles. Realizing that the standard approach used for creating organoids would not work in such a study, the team tried another tactic—they grew stem cells on platform that resulted in a brain organoid that was much thinner and rounder than it would naturally grow—and it was also grown on a form surrounding a narrow space. The end result, the team reports, was a brain organoid that resembled a pita. This configuration allowed the researchers to take images of folds as they developed and to supply nutrients to all the cells since blood vessels typically do not develop in organoids.
In studying the images of the developing organoid, the researchers found that the folds developed as expected—opposing forces resulting from growth differences in brain material. In this case, it was the cytoskeleton in the organoid's core and the cell nucleus expanding at the organoid's outer edges. Uneven expansion between the two causes one or the other to fold as a means of dealing with the increase in pressure.
To learn more about the development of folds, the researchers ran the same experiment again, but used stem cells from a patient with smooth brain syndrome, which, as it sounds, is a condition in which the brain develops without folds. As expected, the organoid developed very few folds. A closer look showed differences in elasticity between the cells in the organoid grown with healthy cells and the those with the mutated genes that are behind smooth brain syndrome.
More information: Eyal Karzbrun et al. Human brain organoids on a chip reveal the physics of folding, Nature Physics (2018). DOI: 10.1038/s41567-018-0046-7
Abstract
Human brain wrinkling has been implicated in neurodevelopmental disorders and yet its origins remain unknown. Polymer gel models suggest that wrinkling emerges spontaneously due to compression forces arising during differential swelling, but these ideas have not been tested in a living system. Here, we report the appearance of surface wrinkles during the in vitro development and self-organization of human brain organoids in a microfabricated compartment that supports in situ imaging over a timescale of weeks. We observe the emergence of convolutions at a critical cell density and maximal nuclear strain, which are indicative of a mechanical instability. We identify two opposing forces contributing to differential growth: cytoskeletal contraction at the organoid core and cell-cycle-dependent nuclear expansion at the organoid perimeter. The wrinkling wavelength exhibits linear scaling with tissue thickness, consistent with balanced bending and stretching energies. Lissencephalic (smooth brain) organoids display reduced convolutions, modified scaling and a reduced elastic modulus. Although the mechanism here does not include the neuronal migration seen in vivo, it models the physics of the folding brain remarkably well. Our on-chip approach offers a means for studying the emergent properties of organoid development, with implications for the embryonic human brain.
Journal information: Nature Physics
© 2018 Phys.org