MXene material could improve sensors that sniff
Sensors that sniff out chemicals in the air to warn us about everything from fires to carbon monoxide to drunk drivers to explosive devices hidden in luggage have improved so much that they can even detect diseases on a person's breath. Researchers from Drexel University and the Korea Advanced Institute of Science and Technology have made a discovery that could make our best "chemical noses" even more sensitive.
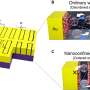
In research, recently published in the American Chemical Society journal ACS Nano, the team describes how a two-dimensional, metallic material called MXene can be used as a highly sensitive detector of gaseous chemicals. The paper suggests that MXene can pick up chemicals, such as ammonia and acetone, which are indicators of ulcers and diabetes, in much lower traces than sensors currently being used in medical diagnostics.
"MXene is one of the most sensitive gas sensors ever reported. This research is significant because it expands the range for detection of common gases allowing us to detect very low concentrations that we were not able to detect before," said Yury Gogotsi, Ph.D., Distinguished University and Bach Professor in Drexel's College of Engineering, who was a lead Drexel author of the study. "The high sensitivity of the device may be used for detecting toxic gases or pollutants found in our environment."
Gogotsi's Nanomaterials Research Group, from Drexel's Department of Materials Science and Engineering, teamed with Hee-Tae Jung, Ph.D., a professor at KAIST in Daejeon, South Korea to explore the gas-sensing properties of titanium carbide MXene. The key to its excellent scent-sleuthing capabilities is that MXene is both highly conductive and undergoes a measurable change of electrical conductivity in the presence of the chemical it's designed to detect—and only when that particular chemical is present.
This discernment is called "signal-to-noise" ratio in the world of chemical sensors and it is used to rank the quality of sensors—picking up more signal and less noise is the goal. The ones in use today—mostly in medical settings to detect chemicals like acetone, ethanol and propanol, or in breathalyzers to detect alcohol—have a signal-to-noise ratio between 3-10, MXene's is between 170 and 350, depending on the chemical.
"If the material can respond to gases by giving a strong signal, while simultaneously being conductive and achieving low electrical noise, the sensor can detect gases at very low concentrations because the signal-to-noise ratio is high—this is clearly the case with MXene," Gogotsi said. "MXene can detect gases in the 50-100 parts per billion ranges, which is below the concentration necessary for current sensors to detect diabetes and a number of other health conditions."
This level of sensitivity could be extremely important for detection of disease. In addition to ulcers and diabetes, breath analysis is currently being developed for early diagnosis of multiple types of cancer, cirrhosis, multiple sclerosis and kidney disease. If the chemical indicators for these diseases can be spotted in lower concentrations they are more likely to be diagnosed and treated at earlier stages.
MXene's advantage over conventional sensor materials lies in its porous structure and chemical composition. The material is good at both allowing gas molecules to move across its surface and snagging, or adsorbing, certain ones that are chemically attracted to it, showing good selectivity.
Gogotsi's team has been exploring MXenes since the material's discovery at Drexel in 2011. The group has been able to create and study more than two dozen different chemical compositions for the material, which means they could be used to create sensors for a very wide variety of gasses.
In the future, Gogotsi suggests, MXene sensors could play an important role in environmental monitoring, energy harvesting and storage, as well as health care.
"The next step to advance this research will be to develop sensor sensitivity to different types of gases and improve the detection selectivity between different gases," Gogotsi said. "We can also imagine personal sensors that will be in our smart phones or fitness trackers, monitoring body functions and the environment while we work, sleep or exercise, accessible with a tap of a finger. Improving the detection sensitivity with new materials is the first step toward making these devices a reality."
More information: Seon Joon Kim et al. Metallic Ti3C2Tx MXene Gas Sensors with Ultrahigh Signal-to-Noise Ratio, ACS Nano (2018). DOI: 10.1021/acsnano.7b07460
Journal information: ACS Nano
Provided by Drexel University