A new method for detecting levels of an important amino acid

A team from the Faculty of Chemistry of MSU and colleagues have suggested a new method for determining levels of cysteine, an amino acid used in many drugs, with the help of gold nanoparticles. Unlike current methods, it does not require complex reactions or expensive equipment. An article with the results of the study was published in Sensors and Actuators B.
Cysteine is an amino acid found in keratins, the proteins comprising nails, hair and feathers. Cysteine deactivates toxins, acts as an antioxidant, protects against X-rays and radiation, and is used in medicinal drugs and food additives. Moreover, its presence in blood may be an indication of certain conditions such as Alzheimer's disease or cardiovascular, liver or skin disorders. Therefore, determining the level of cysteine in biological fluids is a useful diagnostic tool.
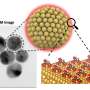
As a rule, the content of cysteine in a solution is detected using the luminescence method, but it requires expensive equipment, additional procedures and qualified personnel. Therefore, a simpler and cheaper procedure is required. The authors of the study suggested using gold nanoparticles that are 20 nanometers in diameter and are simply produced. In a solution, they form a stable colloid system, i.e., they don't sink down to the bottom of the vessel.
"It is very easy to obtain such particles, especially the non-modified ones. We just take a gold compound, say, chloroaudic acid, and sodium citrate (a salt of citric acid), mix them and heat up—that's all the synthesis that is required. The process is simple and can be carried out in almost any lab," said chemist Vladimir Apyari, a co-author of the work, senior research assistant of the Faculty of Chemistry, MSU.
The scientists used gold nanoparticles due to their ability to quickly change color from ruby red to blue upon aggregation into bigger formations. Because of this property, they are used to detect ions of metals, anions and organic compounds. When nanoparticles react with cysteine, they are aggregated, and the solution's spectral characteristics and colour change. These changes can be measured with a spectrophotometer or even seen with a naked eye. The reaction takes a couple of minutes.
Cysteine detection in a solution has certain disadvantages. For example, no spectrophotometer analysis can be carried out in a muddy or coloured environment, and cleansing is required. Instead, the nanoparticles may be placed on the surface of a solid material, such as polyurethane foam. The particles are completely consumed by polyurethane foam in 15 to 20 minutes.
According to the authors, in the future, their study may lead to new detection methodologies. More complicated nanoparticles with added analytical groups (chemical structures that bind with the analyzed substance) could also be used. Therefore, the method may be adapted to detect and determine the levels of other compositions.
More information: Vladimir V. Apyari et al, Label-free gold nanoparticle-based sensing of cysteine: New peculiarities and prospects, Sensors and Actuators B: Chemical (2018). DOI: 10.1016/j.snb.2018.01.118
Provided by Lomonosov Moscow State University