Researchers develop process producing cell-sized lipid vesicles for cell-cell synaptic therapies
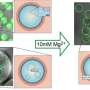
A team of researchers at the University of California in Irvine, California has demonstrated a novel process to produce cell-sized lipid vesicles (CLVs) from microfluidically generated double emulsion templates by investigating the interfacial parameters that control double emulsion stability for storage, and their subsequent dewetting to form multisomes or GUVs (subsets of CLVs). A report detailing this latest development was published in the December 2017 issue of the journal Technology.
Cell-sized lipid vesicles (CLVs) have long been of particular interest for many therapeutic, biosensing, and artificial cell applications, as the lipid bilayer mimics a cell membrane and is a very versatile and selective physical boundary, which can be easily functionalized to meet a variety of needs. This is especially relevant for emerging applications in medicine, where functional particles can be utilized to elicit a therapeutic cellular response, and in biosensing, where small reaction containers can be used to detect even single molecules of a specific target. Unfortunately, the use of CLVs in such areas has been limited to research and laboratory work.
Producing uninform cell-sized, unilamellar lipid vesicles requires microfluidic technology, as common place bulk methods such as gentle hydration and electroswelling typically form heterogeneous populations of unilamellar and multi-lamellar vesicles, with little control over size or material encapsulation. Typical microfluidic methods of producing CLVs eliminate most of these shortcomings, but they are formed from double emulsion templates that utilize volatile solvents that can be detrimental to encapsulated biological materials, and need to be produced on site by trained personnel. By storing them as stable double emulsion precursors, the microfluidic generation does not have to be performed on site, and the CLVs can be formed by the end user for further processing.
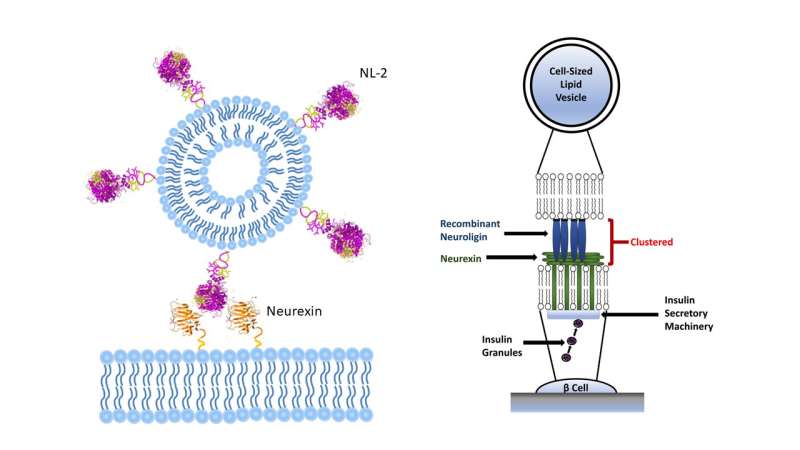
A promising medical application for CLVs involves using them to induce specific cellular responses for a therapeutic effect. The lipid bilayer of the CLVs, much like a cell membrane is a fluid surface that can be functionalized to interact and influence the behavior of other cells through the coordinated arrangement of ligand-receptor pairs into ordered spatial patterns, which is not possible on the surface of solid substrates such as latex or hydrogel beads. Ligand receptor clustering on the cell surface has recently been recognized as a critical aspect that is required in many juxtacrine-signaling pathways. The importance of these fluid micro-domains in cell-to-cell communication is widely recognized in the immune synapse formed when antigen-presenting cells interact with T-cells, and is a prominent design criterion in the creation of artificial antigen presenting cells (aAPCs) for immunotherapy.
To demonstrate the ability of the engineered CLVs to influence cellular behavior, the investigators functionalized them with neuroligin-2 (NL-2), a transmembrane protein in pancreatic β cells that is an important component in the pathway that leads to insulin secretion. Previous studies to induce insulin secretion with monomeric forms of NL-2 in solution have had no success in promoting insulin secretion, and can actually be detrimental to cell survival. By covalently bonding NL-2 to the lipid bilayer surface of CLVs, the investigators were able to induce a 20% increase in insulin secretion in pancreatic β cells compared to blank controls and promoted increased levels of cellular proliferation and growth. Both effects have high therapeutic potential for patients suffering from Type 2 diabetes. These results present a promising platform for these CLVs to be utilized as blank templates for a variety of applications, including cell- therapy that rely on the formation of cell-cell synapses (e.g. artificial antigen presenting cells), drug delivery vehicles and biosensors.
More information: D. Vallejo et al, Cell-sized lipid vesicles for cell-cell synaptic therapies, Technology (2018). DOI: 10.1142/S233954781750011X
Provided by World Scientific Publishing