Carbon monoxide detection in the body could lead to rapid disease diagnostics
A quick and reliable way to detect levels of carbon monoxide in the body could allow clinicians to diagnose disease.
Carbon monoxide is normally considered in terms of the amount of damage it can cause us, but a team of scientists at Imperial College London and the Polytechnic University of Valencia have been looking at the other biological roles it can play.
Carbon monoxide is actually produced in our bodies in small amounts, and is vital in a number of biological processes, often acting as a messenger molecule. With evidence suggesting the involvement of carbon monoxide in resolving inflammation and possibly alleviating cardiovascular disorders, it is a growing area of interest for many scientists.
One exciting possibility is the therapeutic use of carbon monoxide to treat disease, but this, and other related areas, are currently limited by the ability to sensitively track the generation of carbon monoxide in real time.
Now, a new technology, devised by Imperial and Valencia scientists and published in the Journal of the American Chemical Society, has the potential to be used to gauge carbon monoxide levels in the body both effectively and rapidly.
New way to detect carbon monoxide
Initially, the group at Imperial were working on developing colour-changing strips that could indicate carbon monoxide levels in the air around us whilst avoiding interference from steam or smoke. Following the publishing of this research, they started work with their colleagues from Valencia, who were interested in the possible biological applications of the technology.
In order to develop a technique for sensing carbon monoxide in the body, Ph.D. students Anita Toscani and Jonathan Robson had to find ways to detect much smaller concentrations, moving from detecting carbon monoxide in parts per million to parts per billion.
An additional challenge was to achieve this while avoiding unwanted interactions with the many other substances present. The team leader from the Department of Chemistry at Imperial, Dr. James Wilton-Ely, explained that: "The twin challenges are sensitivity and selectivity, and we think we've ticked both boxes."
Glowing signals
Carbon monoxide poisoning occurs when it binds to the iron in our blood's haemoglobin, preventing its ability to carry oxygen from the lungs to the rest of the body. In an interesting twist, this very affinity for certain metals is what makes the new technique so successful. It uses ruthenium (a metal in the same group of the periodic table as iron) with carefully designed organic molecules (ligands) attached.
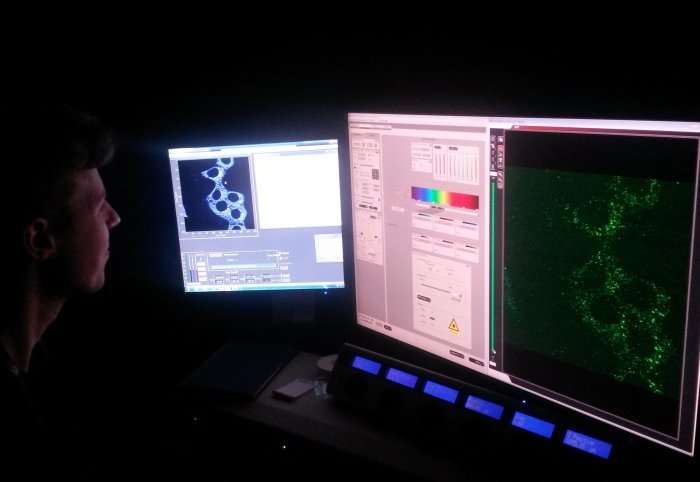
One of these ligands is designed to have the ability to glow, or fluoresce, when light of the correct wavelength is used, but are "switched off" when attached to the ruthenium metal. When carbon monoxide is present, it binds to the ruthenium, detaching the ligand. This causes the ligand to "switch on" and begin to fluoresce under deeply penetrating red light, becoming visible under a microscope.
The researchers were able to test the technique using sample cells taken from mice, and found that they were quickly able to detect the carbon monoxide produced as a response when inflammation was induced in the mice. Using image analysis, they could then quantify the amount of carbon monoxide present from the fluorescence produced.
This technology has great potential for future applications in medical diagnostics, but it also illustrates the different ways we should be thinking about chemicals like carbon monoxide. As Dr. Wilton-Ely pointed out: "This offers a really nice insight into the world around us—so many things have multiple roles and reputations, and this adds a richness to the world of science that should be embraced."
More information: Cristina de la Torre et al. Ex Vivo Tracking of Endogenous CO with a Ruthenium(II) Complex, Journal of the American Chemical Society (2017). DOI: 10.1021/jacs.7b11158
Journal information: Journal of the American Chemical Society
Provided by Imperial College London