The bright side of an infectious protein: Stress sensors promote yeast cell survival
Prions are self-propagating protein aggregates that can be transmitted between cells. The aggregates are associated with human diseases. Indeed, pathological prions cause mad cow disease and in humans Creutzfeldt-Jakob disease. The aggregation of prion-like proteins is also associated with neurodegeneration as in ALS. The regions within prion-like proteins that are responsible for their aggregation were termed prion-like domains. Despite the important role of prion-like domains in human diseases, a physiological function has remained enigmatic. Researchers at the Max Planck Institute of Molecular Cell Biology and Genetics (MPI-CBG), the Biotechnology Center of the TU Dresden (BIOTEC), and the Washington University in St. Louis, USA have now identified for the first time a benign, albeit biologically relevant function of prion domains as protein specific stress sensors that allow cells to adapt to and survive environmental stresses. Uncovering the physiological function is an essential first step towards closing a gap in understanding the biological role of prion domains and their transformation into a pathological disease-causing state.
The discoveries were published in Science.
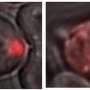
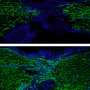
The aggregation of prion-like proteins is associated with human diseases. Their infectious behavior is comparable to the spread of a viral infection. This raises the question of why evolution has kept these proteins around: are these sequences good for anything? In their study, the team around research group leader Prof. Simon Alberti from the MPI-CBG focused on the yeast prion protein Sup35, which has a long-standing history as a role model for prion research. They found that the prion-domain of Sup35 acts like a stress sensor that triggers the formation of protective protein droplets and gels when cells are exposed to harsh conditions.
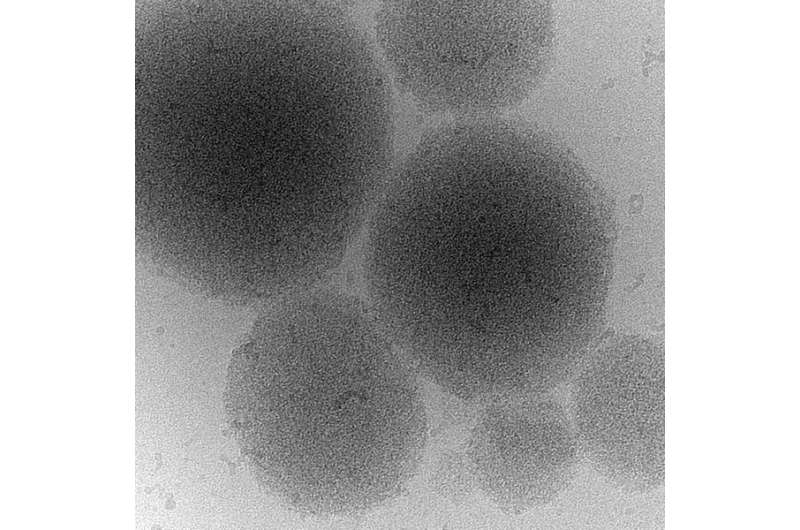
When cells are stressed, for example because they are starved of nutrients, their energy level drops. This leads to a decrease of the cytosolic pH value - the cells acidify. In response, cell division stops, the metabolism shuts down and cells enter into stand-by mode. When the stress is over, cells must rapidly reprogram their metabolism and restart growth and division. Prof. Simon Alberti and his colleagues found out that the Sup35 prion domain is important for stress survival. "We found that cells lacking the prion domain show a growth defect when recovering from stress", summarizes Titus Franzmann, the first author of the study. The scientists discovered that the prion domain of Sup35 senses the acidic pH of the cytosol and then drives the formation of protein droplets that protect Sup35 from damage. "To store the protein the droplets can even advance into a gel-like structure", says co-author Marcus Jahnel from the biophysics group of Prof. Stephan Grill at the BIOTEC. These protein droplets - that form in the cytoplasm similar to condensing water droplets - can dissolve again, enabling the cell to reuse the Sup35 protein when it restarts growth. Additionally, colleagues from the Washington University in St. Louis predicted the sequences of the amino acids responsible for Sup35 sensing changes in the cytoplasmic pH value. In this context, Rohit Pappu, Edwin H. Murty Professor of Biomedical Engineering at Washington University, noted that: "Uncovering the molecular components that confer these adaptive abilities of Sup35 has also important implications for understanding cells on a molecular level and adopting these principles for building synthetic systems".
From an evolutionary standpoint, the Sup35 condensates are really interesting, since they are conserved among distantly related yeast that diverged almost 400 million years ago. This suggests that droplet and gel formation may be an ancestral function of the Sup35 prion domain. Titus Franzmann concludes: "The study suggests that prion domains are protein-specific stress sensors that allow cells to respond to specific environmental conditions. In that way, we could show for the first time a positive function of a prion domain that has often only been associated with disease-causing aggregates. So maybe that's the reason why evolution has kept them for so long."
More information: Phase separation of a yeast prion protein promotes cellular fitness, Science 05 Jan 2018 , DOI: 10.1126/science.aao5654 , science.sciencemag.org/content/359/6371/eaao5654
Journal information: Science
Provided by Max Planck Society