A bacterial powder for quickly stabilizing gravel surfaces

EPFL's Laboratory of Soil Mechanics has developed an easily reproducible technique using bacteria and urea to reinforce sandy or gravelly terrain. A series of chemical reactions lead to the rapid formation of mineral crystals that bind the ground particles together.
Much of the buildable land in urban areas is already in use. That which remains often needs to be stabilized and reinforced in order to minimize long-term environmental risks and the possibility of crack formation. Existing approaches to this problem include injecting concrete or putting down deep foundations, but these techniques are often impractical, unprofitable or environmentally undesirable. Dimitrios Terzis and Lyesse Laloui, two researchers at EPFL's Laboratory of Soil Mechanics (LMS), have come up with an organic, easy-to-use and low-cost solution that combines bacteria and urea. These two substances react to create calcite crystals, which firmly bind the gravel or sand particles together. The research on this promising and long-lasting biocement has just been published in Scientific Reports.
The desired result in just a few days
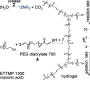
Several chemical reactions are necessary to synthesize calcite crystals. For the triggering agent, the researchers used Sporosarcina pasteurii bacteria, which they freeze-dried for easier handling. When the bacteria are spread over the ground, they bind to sand and gravel particles, reproduce and form a protective adhesive layer. The bacteria then serve as matchmakers between urea, a highly soluble, non-toxic synthetic molecule, and calcium, which are pulverized above ground.
The bacteria break down the urea molecules for use as food and to improve their environment. This releases carbonate, which binds to the calcium to form calcite crystals. The crystals attach to the ground particles and grow in both number and size – in some cases reaching several hundred micrometers across. Urease, an enzyme also released as part of the bacteria's digestive process, accelerates this process significantly – making the reaction 1,000 times faster – and the end product is achieved in a matter of days or even hours. It turns out that once the bacteria have deteriorated, the urease enzyme alone can break down the urea molecules. Because urease is just as effective as the bacteria at doing this, the researchers attempted – successfully – to remove the bacteria from the process. "A 'cell-free' alternative would make it easier to use this method on a very large scale at a lower cost," says Lyesse Laloui, a professor at LMS.
A scalable solution
The resilience of this biocement and its microstructure has been tested on various types of soil, with excellent results. As part of their research, the LMS team also developed a new quantitative observation method. They were able to conduct x-ray microtomography analyses and 3-D reconstructions in order to see the growth mechanism up close and the condition of the crystal bonds. They took particular note of the size, number and orientation of these bonds. This information provided them with a better understanding of how their method works in practice.
The biocement can be produced on site at ambient temperatures through a low-cost process that consumes little energy. In addition, its use can be scaled to the needs of any given construction project. Small quantities of calcite generate enough particle resilience for gravel to withstand the shear stresses caused by a major earthquake; they can also stabilize sloping surfaces or shore up existing foundations. With higher levels of calcite, the biocement can serve as a construction material or be used for soil sealing.
Journal information: Scientific Reports
Provided by Ecole Polytechnique Federale de Lausanne