Two studies find stress reprograms cells
In a pair of publications, researchers have shown how cells adapt to stressors—like water loss—by reprogramming their internal signaling networks. The studies describe previously unknown mechanisms that cells use to send signals between cellular machinery and avoid cell death. According to the authors, drugs that enhance the adaptation mechanisms could help cells stave off multiple diseases, including type 2 diabetes. The studies were published in Cell Reports and Molecular Cell.
"We discovered that cells switch off non-essential functions when faced with threats to their survival. At the same time, cells activate new mechanisms to sustain essential functions," said Maria Hatzoglou, PhD, senior author on both papers, professor of genetics and nutrition at Case Western Reserve University School of Medicine, and member of the Case Comprehensive Cancer Center.
The studies focus on two types of cellular stress: cell shrinkage from water loss, and dysfunction of the endoplasmic reticulum, the cellular organelle that makes and guides proteins to specific destinations. Both have diverse causes—everything from a person's genetics to a cell's environment—and cells must adapt to the stressors, or perish. In the new studies, researchers describe a series of cellular workarounds. "Cells protect themselves by slowing down energy-consuming processes, such as protein synthesis. This prepares cells to reprogram the cellular machinery to make only the essential proteins needed for survival," Hatzoglou said.
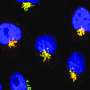
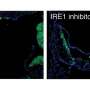
In the Molecular Cell study, Hatzoglou and colleagues describe how stressed cells focus on critical protein production to sustain the function of the endoplasmic reticulum, by transmitting signals along unexpected molecular pathways. In the Cell Reports study, researchers put cornea cells in salt water to dry them out. The cells responded by activating pathways that help transport amino acids. By doing so, the cells were able to prevent water loss. The findings suggest medications that target the transport pathways could help treat dry eye syndrome. Such medications may also help treat neurodegenerative diseases caused by defects in protein equilibriums.
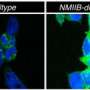
Said Hatzoglou, "Both our papers contribute to our understandings of the ways cells try to ensure proteins are properly folded and navigated through the cells, so they can reach their final destinations and assume their normal functions. This process is known as the 'integrated stress response.'" In Molecular Cell, researchers outline how cells can "reprogram" the integrated stress response. As the journal explains, the authors "unravel the mechanism of adaptation to chronic stress that encompasses previously unappreciated remodeling." The study outlines a cascade of new cellular signals that cells use to adapt to stressful conditions. Interestingly, the novel stress defense mechanism involves remodeling of the cellular machinery that translates mRNA genetic material into a select group of proteins. This selective protein synthesis protects stressed cells from life-threatening endoplasmic reticulum dysfunction.
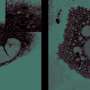
In Molecular Cell, Hatzoglou and coworkers also show that endoplasmic reticulum dysfunction is marked by a novel cell death mechanism involving cytoplasmic vacuolization—the formation of large, toxic pockets inside cells that is reminiscent of foamy cells, observed in many human pathologies. This phenotype is a pathologic feature in the brains of the neurodegenerative diseases called childhood ataxias, and introduces the interesting possibility that their cause is the result of endoplasmic reticulum dysfunction in nerve cells.
The findings could also lead to new diabetes treatments. "Patients who develop type 2 diabetes become ill because they make too much insulin. This causes the endoplasmic reticulum to be overwhelmed and unable to handle the sudden protein overload, leading to dysfunction. This dysfunction later kills insulin-producing cells in the pancreas," said Hatzoglou. "We believe by enhancing the adaptive response to increased insulin we can delay endoplasmic reticulum dysfunction and the onset of disease." Hatzoglou received funding from the National Institute of Diabetes and Digestive and Kidney Diseases to study how activating cellular stress responses could help delay diabetes progression.
Together with colleagues, Hatzoglou is planning future experiments to understand the molecular mechanisms that defend cells during diverse stress conditions. The findings could lead to new therapeutics to prevent cell death in multiple disease states—from dry eye syndrome to diabetes.
More information: Dawid Krokowski et al, GADD34 Function in Protein Trafficking Promotes Adaptation to Hyperosmotic Stress in Human Corneal Cells, Cell Reports (2017). DOI: 10.1016/j.celrep.2017.11.027
Bo-Jhih Guan et al. A Unique ISR Program Determines Cellular Responses to Chronic Stress, Molecular Cell (2017). DOI: 10.1016/j.molcel.2017.11.007
Journal information: Cell Reports , Molecular Cell
Provided by Case Western Reserve University