Researchers show how insect food choice can be manipulated
Researchers at the University of California, Riverside have found a way to access and manipulate taste neurons in the pharynx (throat) of the common fruit fly that could help control the spread of mosquito-related illnesses, such as dengue, malaria, yellow fever, and Zika virus, and reduce the loss of crops due to agricultural pests.
In insects, taste receptors are found in neurons present in external taste hairs on wings, legs and mouthparts, as well as in internal taste organs in the pharynx. Most researchers studying taste focus only on the external taste hairs, however, and overlook the pharynx, which is harder to access.
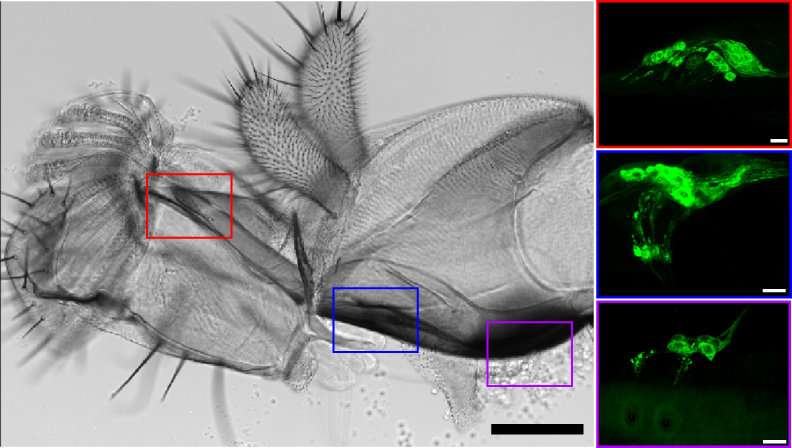
Anupama Dahanukar, an associate professor of molecular, cell, and systems biology, and Yu-Chieh David Chen, her graduate student, were able to examine the molecular identities of pharyngeal neurons in the fruit fly, a powerful genetic model organism for studying insect behaviors, by making the neurons fluorescent. The fluorescence helped them understand, at a molecular level, how the pharyngeal neurons were organized.
"Using transgenic flies, we were able to examine the expression of fluorescent proteins designed to reflect patterns of chemosensory receptors in pharyngeal taste neurons in the fly and draw a precise molecular map of these organs, which has not been done before," said Dahanukar, who led the research project. "We were also able to manipulate selected groups of these neurons to figure out whether they inform the fly to eat certain foods or to avoid them."
Such genetic manipulation of neurons in insects could have vast applications in curbing the spread of mosquito-borne diseases and reducing crop damage by controlling the feeding behavior of mosquitoes and agricultural pests, respectively. Malaria alone killed about 438,000 people in the world in 2015; approximately, 3-4 billion people are at risk of contracting dengue or malaria each year. Losses to U.S. crop and forest production are estimated at $40 billion annually.
"The important role that a fly's pharyngeal taste organs play in regulating food choice has gone largely unappreciated until recently," said Michael Gordon, an associate professor of biology at the University of British Columbia, who was not involved in the research. "Anupama and David's very careful and detailed mapping of specific pharyngeal cell types offers important insights into the logic of chemical detection in these organs. It also provides a framework for further understanding the complexities of taste's impact on fly feeding behaviors."
Study results appear in Cell Reports.
"The genetic toolkit we have defined can allow us to manipulate selected classes of pharyngeal taste neurons and examine consequences of these manipulations of various functions," Chen said.
Chen and Dahanukar also found that the fly's pharyngeal taste organs share some features with taste hairs on the external organs. Other features, such as co-expression (or lack thereof) of certain receptors, may be unique to pharyngeal organs.
"It has been suspected that information from pharyngeal organs might be interpreted differently from information coming from external organs, because neurons from different organs connect to different circuits in the brain," said Dahanukar, a member of the Institute for Integrative Genome Biology at UCR. "But molecular differences between the two suggest that the nature of the information might also be different. Pharyngeal organs may sample chemicals differently from external organs or these internal neurons may sense other food attributes, which help the fly decide whether or not to ingest it."

To achieve fluorescence in the fly's pharyngeal taste neurons, Dahanukar and Chen mated two transgenic flies, with one parent fly having a GAL4 transgene under the control of regulatory regions of different chemosensory receptors and the other parent having a UAS-GFP transgene. When the two transgenic flies mated, the offspring's pharyngeal taste neurons showed fluorescence in patterns that reflect expression of those chemosensory receptors.
Chen explained that the UAS-GFP transgene allows expression of the green fluorescence protein, but it cannot achieve this on its own. To generate fluorescence, a chemoreceptor-GAL4 is needed to turn on the genes downstream of the UAS sequence.
"The reason we don't introduce both GAL4 and UAS transgenes in the same fly but instead separate the GAL4 and UAS transgenes into different transgenic fly lines is that we want to make these fly lines more versatile for combinatorial experiments," he said. "Consider capsaicin, an active component of chili peppers that flies cannot taste. If we can express a capsaicin receptor in a pharyngeal neuron of the fly using a particular combination of chemosensory-GAL4 and UAS-capsaicin transgenes, then that neuron now has the receptor for capsaicin. This means capsaicin can now activate this neuron. In this way, we can give the fly any taste receptor we want and alter the fly's behavior. By artificially activating selective neurons, we can make the fly approach a certain food or move away from it."
Based on their location in the food canal, pharyngeal taste organs have been thought to be important for controlling feeding behaviors. More and more studies indicate that pharyngeal taste neurons and circuits may be key players in feeding, hunger, reward, and memory.
"It's crucial to study these neurons, which are often bypassed in previous research, because we know food must pass through the pharynx," Chen said. "Focusing only on the external taste organs, such as the legs and wings, could lead to incorrect or incomplete conclusions. Our paper provides road maps of pharyngeal taste organs in an insect model system for probing the role of these understudied neurons in controlling feeding behaviors."
Now that Dahanukar and Chen have genetic toolkits to manipulate fly neurons, they plan to study individual pharyngeal neurons to fully understand what role they play in the fly's feeding behavior.
Journal information: Cell Reports
Provided by University of California - Riverside