Microbial resident enables beetles to feed on a leafy diet

An international team including researchers from the Max Planck Institute for Chemical Ecology has described a bacterium residing in a species of leaf beetles which has an unexpected feature: it provides the beetle with the enzymes required to break down certain plant cell wall components. The genome of the bacterium is the smallest ever sequenced of any organism living outside a host cell. It contains genes that are responsible for the production of pectinases, the enzymes that break down pectin, an essential component of the plant cell wall. The production of pectinases is therefore the primary function of these bacteria. Without bacterial symbionts the beetles could not to gain access to the nutrients inside the plant cells and hence would be unable to survive. The study is reported today in Cell.
"The bedrock of our study were the histological descriptions and drawings of the beetle's symbiotic organs that were first published by the German zoologist Hans-Jürgen Stammer more than 80 years ago. We wanted to molecularly characterize the extraordinary partnership between a leaf beetle and its symbionts described by Stammer as a rare peculiarity," first author Hassan Salem, formerly a doctoral student at the Max Planck Institute for Chemical Ecology, explains. He is currently a Feodor Lynen Postdoctoral Fellow of the Alexander von Humboldt Foundation at Emory University in Atlanta, Georgia, USA.
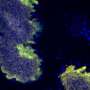
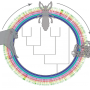
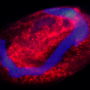
Hans-Jürgen Stammer (1899-1968) studied symbiotic partnerships between insects and bacteria during the 1920s and 30s. He found that leaf beetles (Chrysomelidae) are notorious for not partaking in symbiotic partnerships. However, his studies also revealed that there were exceptions to this finding among some tortoise beetle species, such as the thistle tortoise beetle (Cassida rubiginosa). These tortoise beetles are equipped with unusual organs, which he described in a 1936 study. The symbiotic bacteria reside in sac-like reservoirs in the guts of the beetles. Female beetles transfer the symbionts through vaginal tubes to their offspring by applying a tiny symbiont caplet to each egg. The hatching larvae eat through the egg shell and then consume the caplets containing the symbiotic bacteria.
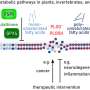
Leaf beetles are able to degrade components of the plant cell wall, such as cellulose and pectin, with the help of digestive enzymes. However, genetic analysis showed that the thistle tortoise beetle lacks the genes that are responsible for the production of the respective enzymes (pectinases). The new study reveals that this deficit is compensated for by a close partnership with a bacterium that resides in special organs close to the beetle's gut. To understand the importance of the bacterial symbionts for the beetle, the authors conducted a range of bioassays, some that were combined with enzymatic measurements. "When we compared enzyme activity in tortoise beetles with and without symbiotic bacteria, we found that beetles without symbionts were not able to digest pectin in order to gain access to the nutrients in the cell and as a consequence their chances of survival decreased," says Roy Kirsch from the Max Planck Institute for Chemical Ecology.
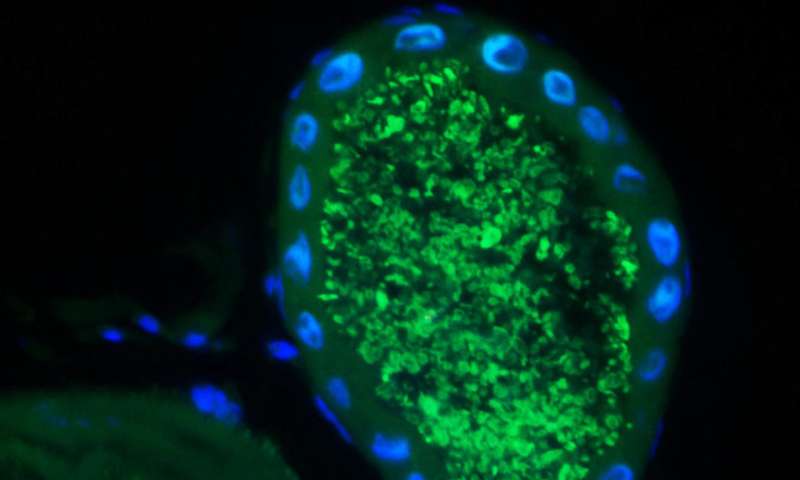
Genetic analysis of the symbiotic bacteria which the researchers present as "Candidatus Stammera capleta" in their current study to honor Stammer's observations uncovered another surprise: The genome of the microorganism is reduced to only a few hundred genes, among them some genes that regulate the production and transport of pectinases. Accordingly, the genome is tiny: Containing only ~270.000 base pairs, the genome of the bacterium is the smallest ever described for an organism that exists outside a host cell. Escherichia coli bacteria in comparisonbacteria that live inside the gut of many animals including humans have 4.600.000, or 17 times as many base pairs. Only some bacteria that are housed inside their hosts' cells are known to have smaller genomes than the beetle symbiont.
The symbiosis between the beetle and its symbiont is characterized by a clear division of labor. "The beetle host possesses the genes responsible for producing cellulases to digest cellulose, whereas the symbiont provides the pectinases. Together they have the necessary enzymes to break down the plant cell wall. Especially notable is the fact that this is the first description of a specialized bacterial symbiont with a primary or even sole function dedicated to pectin degradation," Hassan Salem summarizes.
That many herbivorous animals exist today is the result of adaptations that evolved over time. In fact, microorganisms have played a major role in many such adaptations. The thistle tortoise beetle is an impressive example. Without pectinases it would have no access to nutrients inside a plant cell. The production of these enzymes has been outsourced to a service provider: a bacterium living in special organs near its gut.
In many leaf beetle species, the genes that activate digestive enzymes for the degradation of plant cell walls originated from fungi and bacteria and were introduced into the genomes of the beetles' ancestors via horizontal gene transfer. "It is fascinating that insects have solved the problem of how to break up plant cell walls so differently. Why some insects acquired genes from microbes horizontally, while others maintain symbionts to do the same job is an interesting question that remains to be answered in future studies," says Martin Kaltenpoth from the University of Mainz.
More information: Salem, H., Bauer, E., Kirsch, R., Berasategui, A., Cripps, M., Weiss, B., Kogy, R., Fukumori, K., Vogel, H., Fukatsu, T., Kaltenpoth, M. (2017). Drastic genome reduction in an herbivore's pectinolytic symbiont. Cell 172, DOI: 10.1016/j.cell.2017.10.029
Journal information: Cell
Provided by Max Planck Society