Ice skating on water, even when it is really cold
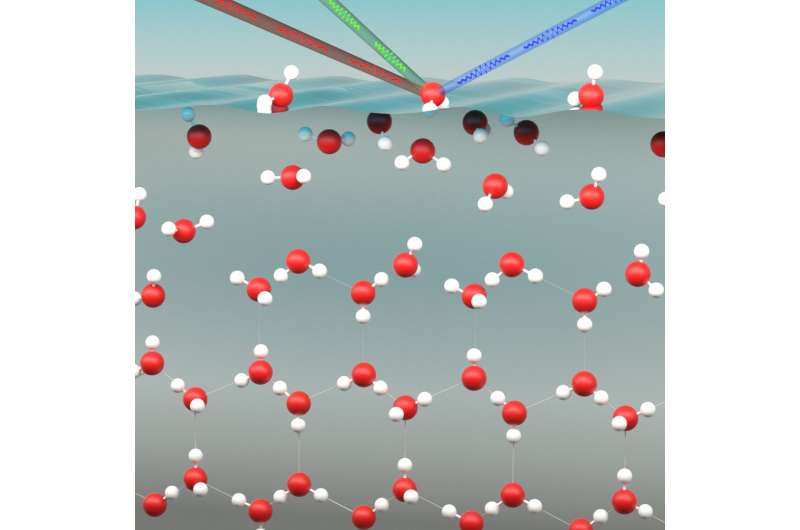
The outermost layer of ice behaves like liquid water, even at a temperature of –30°C. Physicists at AMOLF have irrefutably demonstrated this using a modern surface-sensitive measuring technique. At lower temperatures, however, the layer of water becomes increasingly thin. The researchers report their findings in the journal Angewandte Chemie.
One of the reasons ice is so slippery is that the outermost layer is more similar to a liquid than a solid. Researchers from Amsterdam have now experimentally demonstrated that the surface of ice has the same characteristics as liquid water, even at –30°C. This thin layer of water also explains why two ice cubes can freeze together when they come into contact, which does not happen with other materials.
AMOLF researchers Wilbert Smit and Huib Bakker studied the strength of the bonds between water molecules in the top layer of ice. As the surface is very thin, they used a sensitive technique that can visualize the behavior of only the outermost molecules of the surface. In previous efforts, the measuring equipment could not distinguish between the top layer and the rest of the ice.
The two researchers found that the liquid outermost layer became increasingly thin as the temperature dropped, from four molecular layers at –3°C to two molecular layers at –30°C. As the ice is cooled further, even the outermost layer eventually becomes frozen. That is one of the reasons that ice becomes less slippery at temperatures below –30°C. In those circumstances, ice skating becomes increasingly more difficult.
The researchers used an advanced technique called sum-frequency generation spectroscopy. This technique makes it possible to register the behavior of the surface very specifically, without passing on any information about the area underneath. If the surface is illuminated with two intense light beams of very rapid (femtosecond) lasers, then, under the right conditions, the two light beams only interact with the molecules on the surface. This produces a light beam of a different color. The color and intensity of this beam contain detailed information about the molecular structure of the surface.
More information: Wilbert J. Smit en Huib J. Bakker, The Surface of Ice is Like Supercooled Liquid Water, Angewandte Chemie, 27.11.17, DOI: 10.1002/anie.201707530
Journal information: Angewandte Chemie
Provided by AMOLF





















