A huge hydrogen generator at the Earth's core-mantle boundary
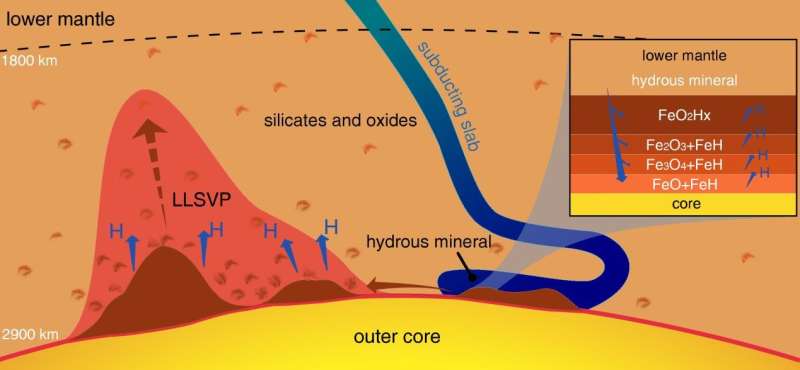
Splitting water into hydrogen and oxygen is a major theme in the development of clean, abundant energy source. A new study led by an international research group revealed that when water meets the iron core of the Earth, the extremely high pressures and temperatures existing at the core-mantle boundary can naturally cause water to split into hydrogen and a super oxidized iron dioxide. Both the released hydrogen and the retained oxygen in the dioxide have many far-reaching implications and consequences, including the behaviors of the core-mantle boundary as a huge hydrogen generator, the separation of the deep Earth's water and hydrogen cycles, and the accumulation of oxygen-rich patches.
The article, published in the National Science Review, is the result of an international collaboration among the Center for High Pressure Science and Technology Advanced Research (HPSTAR) in China and Carnegie Institution for Science in Washington, DC, and Department of Geosciences, Stanford University. They conducted high pressure-temperature experimental studies and theoretical calculations on the reaction between water and iron and probed the reaction products with synchrotron X-ray sources at the Advanced Photon Source, Argonne National Laboratory. They observed a series of intermediate composition iron oxides and iron hydride, with the final product of hydrogen and the new super oxidized iron dioxide.
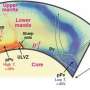
The authors argue that, based on our knowledge of water in the slabs subducting into the deep interior as a result of plate tectonics movement, 300 million tons of water per year could be carried down and meet iron in the core. This could generate a great amount of free hydrogen at the core-mantle boundary 2900 kilometers beneath the surface. Such a rich hydrogen source is far beyond our reach. But understanding the geochemistry of deep volatiles will involve studying the return of hydrogen to the surface as free hydrogen, as carbon hydrides through reactions with carbon, as hydrides through reaction with nitrogen, sulfur, and halogens, or as water after recombining with oxygen on the way up.
Furthermore, the authors point out that continuous accumulation of super-oxidized iron dioxide at the core-mantle boundary throughout the Earth's history may create sizable domains detectable by seismic probes. Such domains may stay at the core-mantle boundary indefinitely without disturbance. However, they are out of place in terms of their highly oxidized chemistry in the reduced environment near the iron core. In the event of overheating by the core, a massive amount of oxygen could be released and erupt to the surface, causing a colossal episode such as the Great Oxidation Event 2.4 billion years ago, which pumped oxygen into the atmosphere and enabled aerobic life to develop.
"This newly discovered water-splitting reaction at the middle Earth affects geochemistry from the atmosphere to the deep interior," said the lead-author Ho-kwang Mao. "Many previous theories need to be re-examined now."
More information: Ho-Kwang Mao et al, When water meets iron at Earth's core–mantle boundary, National Science Review (2017). DOI: 10.1093/nsr/nwx109
Provided by Science China Press