Cryo-EM imaging suggests how the double helix separates during replication
Life would be impossible if the DNA in dividing cells were replicated with anything less than near-perfect precision. Every time a nucleated cell commits to becoming two cells, every "letter" of its genome must be replicated once and only once. In humans, the task boggles the imagination. If unwound, the double helix crammed into each of our cells would measure 6 feet in length. In our bone marrow alone, half a billion new cells are born every minute. These cells alone contain enough DNA to wrap around the earth's equator 25 times. Within daunting tolerances, each new cell must have a genome identical to that of the cell that gave birth to it. Cancer and other diseases can result when the process goes awry.
Figuring out how accurate replication works at the level of individual molecules and atoms is one of the great achievements of modern science. The journey of investigators is not yet done, however. A major unsolved part of the puzzle is understanding how the entire process of copying the genome begins. In new research, insight into how the two stands of the double helix separate in the earliest stages of replication is becoming clear.
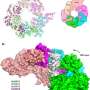
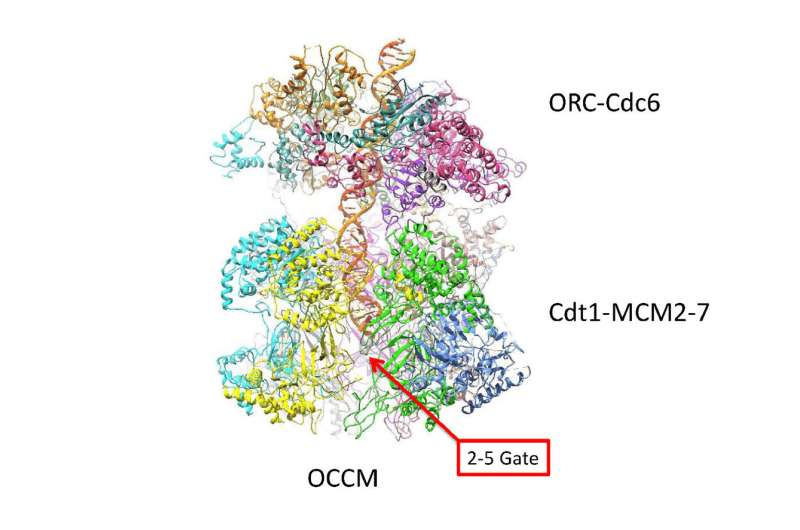
A longstanding collaboration by researchers in London, Grand Rapids, Michigan and Cold Spring Harbor Laboratory (CSHL) in New York reports the atomic-level structure of twin helicase enzymes loaded head to head, with the DNA double helix visible in the circular channel that runs though both helicases. The configuration, a part of the pre-replicative complex (pre-RC), has never been successfully imaged in this configuration before.
The feat was made possible by a new cyro-electron microscopy (cryo-EM) facility at the Van Andel Research Institute, home of one of the lead investigators, Dr. Huilin Li. Dr. Li has collaborated with Dr. Bruce Stillman of CSHL and Dr. Christian Speck, Professor of Genome Biochemistry and Molecular Biology at Imperial College in London for over a dozen years. In 1992, Stillman and colleagues discovered the protein complex called the origin replication complex (ORC), which assembles protein complexes at many locations called "start sites" along the double helix, where replication initiates. Dr. Speck's work showed that ORC combines with other proteins—Cdc6, Cdt1 and the hexamer of Mcm2-7—to begin the process of duplicating DNA.
A great deal of past effort has revealed how ORC assembles and finds start sites. There are many such sites, organized by domains, in the complex human genome; many fewer in simpler forms of life such as baker's yeast. The new research concerns what happens after the initial recognition of the start sites and how the DNA helix might be unwound.
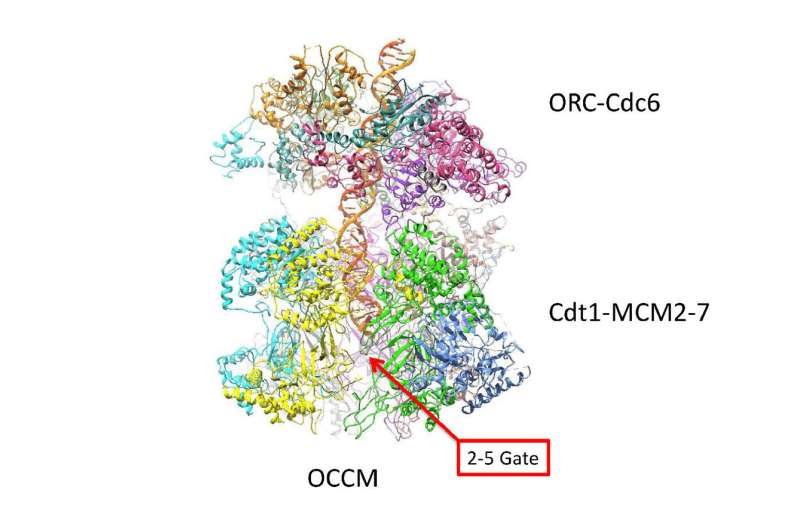
As vividly shown in the new cryo-EM "pictures," the twin six-sided Mcm2-7 helicase enzymes that surround the double helix look like symmetrical insects or, perhaps, twin spacecraft docked head to head. The question answered by the new structure is how the double helix is situated within the channel they form, and how DNA interacts with the surrounding structure. Based on that new knowledge, insight into how the two DNA strands separate, long a mystery, is beginning to be uncovered.
"The new images show that once loaded into the double hexamer—or DH, as we call the head-to-head helicases - the double helix takes a zig-zagging path through the central channel, which is sort of kinked," the authors explain. "The two barrel-shaped hexamers are poised in such a manner that they are ready to untwist the double helix when activated."
One consequence is especially important: the twist in the structure of the complex formed by the double rings creates a tortional strain: they load with an inherent tension that makes them something like a coiled spring. Details in the structure not previously seen reveal how various protein subunits of the twin hexamers latch on to the double helix, via tiny loop-like structures.
The scenario posited by Li, Speck, Stillman and their colleagues is that the twin hexamers load in tension, causing one of the two strands of the DNA passing through them to literally bunch up against a closed "door" on one side of the ring, and the other strand against another closed "door" on the opposite side. The team proposes that one of the two doors springs open when the replication process in activated (through the intervention of protein kinases and other helper molecules).
Through the open door in the helicase - but only on one side - one strand of the double helix is forced out, or "extruded." The team proposes that it becomes what is called the "lagging strand" in the DNA replication process. The other strand, remaining in the center of the helical channel, becomes the "leading strand" in replication. Molecular motors loaded onto the two hexamers provide energy for their separation. One activated helicase passes the other, as replication of each strand proceeds in opposite directions, as deduced by biologists decades ago.
The latest structures were made possible by advances in the technique called cryo-electron microscopy, where an electron beam is passed through frozen, single protein-DNA particles to obtain a near atomic-level 3-dimensional image. The key developers of the method, which is now widely used, received the 2017 Nobel Prize for Chemistry.
More information: Yasunori Noguchi el al., "Cryo-EM structure of Mcm2-7 double hexamer on DNA suggests a lagging-strand DNA extrusion model," PNAS (2017). www.pnas.org/cgi/doi/10.1073/pnas.1712537114
Journal information: Proceedings of the National Academy of Sciences
Provided by Cold Spring Harbor Laboratory