How animal genes go into battle to dominate their offspring

The burdens of becoming parents are often shared unequally between male and female animals. This is particularly true of species that give birth to live young, where male duties such as defending the breeding territory and building dens or nests rarely compare with the ordeals of pregnancy and labour.
You might have thought that animals just "accept" this imbalance and get on with it. But actually, they compete over how much each parent contributes. This isn't like the competition to win a mate, with locking horns or displays of plumage. Instead this remarkable battle takes place at the level of the genes.
It now appears it may have evolved very early in animal evolution, perhaps among the first child-bearing animals. What is more, it may even help to explain why animals diversified into different lineages.
Creatures great and small
One arena in which this battle plays out is over the size of offspring. In principle it's in both a mother's and father's interests to produce bigger newborns, since they are more likely to prevail in the struggle for food and survival.
Yet live-bearing females are more likely to die giving birth to larger offspring or become unable to reproduce again. Their mates needn't care – unless they are likely to sire more broods together, as with humans and certain gibbons, wolves and mice. Otherwise, the males' only concern is that their mate invests as much as possible in the offspring they produce together.
This common conflict of interests manifests itself in various ways in nature. Males often desert pregnant females – from birds to humans, for example – thereby leaving them with the burden of bringing up the young. More rarely, in some normally biparental species females desert males. We see this in some beetles, for example.
The genetic battle mentioned previously is another manifestation of this conflict. The males of many species can manipulate the genes that they pass on to their offspring so that they induce extra growth at the expense of the mother. As with desertion, this effectively hands the female a greater share of the child-bearing burden than is in her interests.
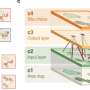
It works as follows. When an embryo grows inside its mother, it consumes resources from her, signalling its metabolic needs along the way. These signals are influenced by certain hormones which either come from the growth genes of the mother or father. The males manipulate the females to deliver more resources by increasing the extent to which these hormones are produced through a chemical modification of their growth genes during sperm formation.
Females have evolved mechanisms to resist this. They can, for instance, pass on to their offspring what is known as a "silenced copy" of their own growth gene. This can counterbalance the male genes' influence by making the embryo grow less than it otherwise would.
This battle is far less prevalent in truly monogamous species, including humans. This goes back to the fact that it becomes less genetically necessary where the two parents have a common interest in the female producing more offspring in future.
Mouse control
British microbiologist David Haig first proposed in 2003 that this battle was more likely in organisms where one sex disproportionately contributes to the offspring, such as live-bearing species, particularly polygamous ones. This was used to explain the puzzling size of the offspring of crosses between oldfield mice and deer mice.
Separately, these species produce similar sized offspring. Yet crosses between male deer mice and female oldfield mice produce offspring that are larger, while the offspring from female deer mice and oldfield males are smaller. Oldfield mice are monogamous while deer mice are polyandrous, meaning one female mates with several males.

Mimicking nature by artificially manipulating a growth gene called igf2, researchers showed that these smaller and larger offspring were due to genetics. In further support of the theory, placental mammals and marsupials including kangaroos and opossums have since been found to have signs of female resistance to such male manipulation.
How early did this mechanism evolve? Researchers have previously suggested it arose in live-born mammals, and would therefore be absent in egg-laying mammals – such as the platypus – and other vertibrates.
But that raises questions about all the reptiles, amphibians and fish which produce live young, since the same genetic manipulation would equally be in their males' interests. To see if it was present, we looked at a Mexican fish called the amarillo or dark-edged splitfin (see lead image).
Along with co-researchers Yolitzi Saldívar and Jean Philippe Vielle Calzada, we crossed males and females from two distant populations of these fish, since they would not have evolved mechanisms which cancel one another out in the way that a single population is likely to have. Sure enough, the size of the embryos was influenced by the specific combination of father and mother. We found signs of male manipulation and probable resistance from the females.
Though based on a small sample size, this suggests that these mechanisms evolved much earlier than previously believed: fish split from other vertebrates some 200m years before live-bearing mammals appeared, dating back about 370m years in total. Whether it comes from a single evolution or from several in different lineages, we cannot yet tell.
One consequence of these genetic battles is the effect on reproductive compatibility within a species. The genetic mutations aimed at manipulating offspring that take place among males and females within a certain group of a species are like a sort of arms race. The genes continually adapt and counter-adapt to one another to try and further their reproductive interests.
If they then mate with an animal from a different group of the same species, their genetic mutations can have made them sufficiently unmatched over time that they are unable to reproduce – thus they are now two species. If this started happening much earlier in evolution than was previously thought, it is likely to have influenced how different groups of live-born animals diverged, including lizards, sharks and mammals. From little acorns, these are the kinds of big oak trees that can grow.
Provided by The Conversation
This article was originally published on The Conversation. Read the original article.![]()