Carbon displays quantum effects

Chemists at Ruhr-Universität Bochum have found evidence that carbon atoms cannot only behave like particles but also like waves. This quantum-mechanical property is well-known for light particles such as electrons or hydrogen atoms. However, researchers have only rarely observed the wave-particle duality for heavy atoms, such as carbon. The team led by Prof Dr Wolfram Sander and Tim Schleif from the Chair for Organic Chemistry II together with Prof Dr Weston Thatcher Borden, University of North Texas, reports in the journal Angewandte Chemie.
"Our result is one of few examples showing that carbon atoms can display quantum effects," says Sander. Specifically, the researchers observed that carbon atoms can tunnel. They thus overcome an energetic barrier, although they do not actually possess enough energy to do that.
Rarely observed for heavy particles
Wolfram Sander explains the paradox: "It's as though a tiger has left his cage without jumping over the fence, which is much too high for him. But he still gets out." This is only possible if he behaves like a wave, but not if he behaves like a particle. The probability of an object being able to tunnel depends on its mass. The phenomenon can, for instance, be observed much more easily for light electrons than for relatively heavy carbon atoms.
The researchers investigated the tunnel reaction using the Cope rearrangement, a chemical reaction that has been known for almost 80 years. The starting material for the reaction, a hydrocarbon compound, is identical to the product molecule. The same chemical compound thus exists before and after the reaction. However, the bonds between the carbon atoms change during the process.
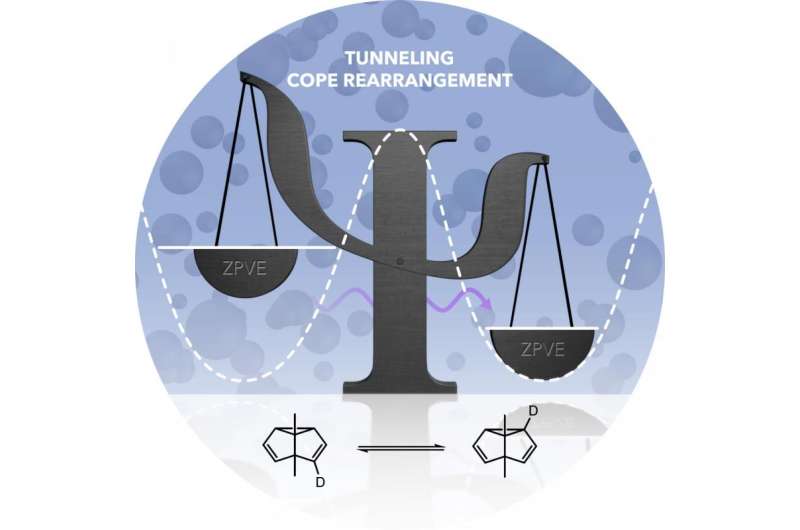
In their experiment, the Bochum-based researchers marked one of the carbon atoms in the molecule: They replaced the hydrogen atom bonded to it with the hydrogen isotope deuterium, a heavier version of hydrogen. Molecules before and after the Cope rearrangement differed in terms of the distribution of the deuterium. Due to these different distributions, both molecular forms had slightly different energies.
Reaction shouldn't actually take place
At room temperature, this difference has little effect; due to the plentiful supply of thermal energy in the surrounding area, both forms occur equally frequently. However, at very low temperatures under ten Kelvin, one molecule form is significantly preferred due to the energy difference. When transitioning from room temperature to extremely low temperatures, the balance has to move from an equal distribution of both forms to an uneven distribution.
This transition cannot, however, occur in the classic way – since, when rearranging from one form to the other, an energy barrier has to be overcome, although the molecule itself does not have the energy for this and the cold environment is also unable to provide it. Although the new balance should not occur in the classic way, the researchers were nevertheless able to demonstrate it in the experiment. Their conclusion: the Cope rearrangement at extremely low temperatures can only be explained by a tunnel effect. They thus provided experimental evidence for a prediction made by Weston Borden over five years ago based on theoretical studies.
Solvents influence ability to tunnel
At Ruhr-Universität, Wolfram Sander undertakes research in the cluster of excellence Ruhr Explores Solvation, where he concerns himself with the interactions of solvents and dissolved molecules. "It is known that solvents influence the ability to tunnel," says the chemist. "However, so far it has not been understood how they do that."
More information: Tim Schleif et al. The Cope Rearrangement of 1,5 Dimethylsemibullvalene-2(4)-d1: Experimental Evidence for Heavy Atom Tunneling, Angewandte Chemie (2017). DOI: 10.1002/ange.201704787
Journal information: Angewandte Chemie
Provided by Ruhr-Universitaet-Bochum