Freezing lithium batteries may make them safer and bendable
Yuan Yang, assistant professor of materials science and engineering at Columbia Engineering, has developed a new method that could lead to lithium batteries that are safer, have longer battery life, and are bendable, providing new possibilities such as flexible smartphones. His new technique uses ice-templating to control the structure of the solid electrolyte for lithium batteries that are used in portable electronics, electric vehicles, and grid-level energy storage. The study is published online April 24 in Nano Letters.
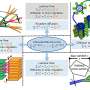
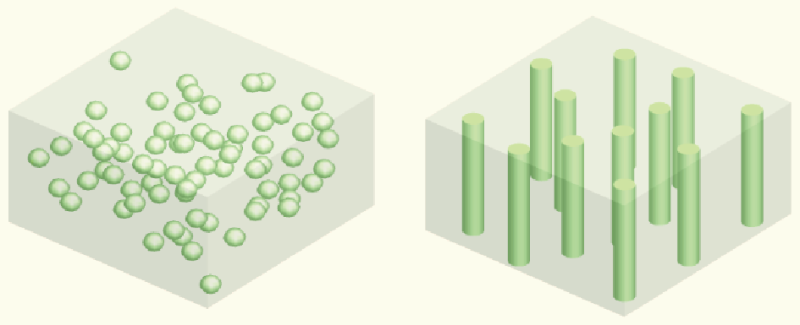
Liquid electrolyte is currently used in commercial lithium batteries, and, as everyone is now aware, it is highly flammable, causing safety issues with some laptops and other electronic devices. Yang's team explored the idea of using solid electrolyte as a substitute for the liquid electrolyte to make all-solid-state lithium batteries. They were interested in using ice-templating to fabricate vertically aligned structures of ceramic solid electrolytes, which provide fast lithium ion pathways and are highly conductive. They cooled the aqueous solution with ceramic particles from the bottom and then let ice grow and push away and concentrate the ceramic particles. They then applied a vacuum to transition the solid ice to a gas, leaving a vertically aligned structure. Finally, they combined this ceramic structure with polymer to provide mechanical support and flexibility to the electrolyte.
"In portable electronic devices, as well as electric vehicles, flexible all-solid-state lithium batteries not only solve the safety issues, but they may also increase battery energy density for transportation and storage. And they show great promise in creating bendable devices," says Yang, whose research group is focused on electrochemical energy storage and conversion and thermal energy management.
Researchers in earlier studies used either randomly dispersed ceramic particles in polymer electrolyte or fiber-like ceramic electrolytes that are not vertically aligned. "We thought that if we combined the vertically aligned structure of the ceramic electrolyte with the polymer electrolyte, we would be able to provide a fast highway for lithium ions and thus enhance the conductivity," says Haowei Zhai, Yang's PhD student and the paper's lead author. "We believe this is the first time anyone has used the ice-templating method to make flexible solid electrolyte, which is nonflammable and nontoxic, in lithium batteries. This opens a new approach to optimize ion conduction for next-generation rechargeable batteries."
In addition, the researchers say, this technique could in principle improve the energy density of batteries: By using the solid electrolyte, the lithium battery's negative electrode, currently a graphite layer, could be replaced by lithium metal, and this could improve the battery's specific energy by 60% to 70%. Yang and Zhai plan next to work on optimizing the qualities of the combined electrolyte and assembling the flexible solid electrolyte together with battery electrodes to construct a prototype of a full lithium battery.
"This is a clever idea," says Hailiang Wang, assistant professor of chemistry at Yale University. "The rationally designed structure really helps enhance the performance of composite electrolyte. I think that this is a promising approach."
More information: Haowei Zhai et al, A Flexible Solid Composite Electrolyte with Vertically Aligned and Connected Ion-Conducting Nanoparticles for Lithium Batteries, Nano Letters (2017). DOI: 10.1021/acs.nanolett.7b00715
Journal information: Nano Letters