How to monitor urine in pools—by testing sweetness
Even though Olympic swimmers have admitted doing it, peeing in the pool is not a condoned practice. Urine contributes to the formation of compounds in pool water that can be harmful to people's health. Now scientists are tackling a new way to monitor water quality: by measuring how sweet it is. Their report appears in ACS' journal Environmental Science & Technology Letters.
Recent studies have shown that nitrogenous compounds (e.g., urea) in urine and sweat react with chlorine to form disinfection byproducts (DBPs), including trichloramine, that can cause eye irritation and respiratory problems. Xing-Fang Li, Lindsay K. Jmaiff Blackstock and colleagues say this evidence has highlighted the need for improved understanding of pool chemistry to raise awareness and educate the public on the importance of swimming hygiene practices. To estimate how much urine—and potentially DBPs—might be in a given pool, Li's team needed to identify what compound might consistently be present in urine. So the researchers turned to the artificial sweetener, acesulfame potassium (ACE), which is marketed as Sunett and Sweet One. The sweetener, which is often used in processed foods like sodas, baked goods and even in other sweeteners, is widely consumed, chemically stable and passes right through the digestive tract and into consumers' urine.
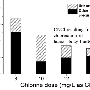
The researchers developed a rapid, high-throughput analytical technique to test more than 250 water samples from 31 actively used pools and hot tubs in two Canadian cities, and more than 90 samples of clean tap water used to initially fill the basins. The concentration of ACE in the pools and hot tubs ranged from 30 to 7,110 nanograms per liter of water—up to 570 times more than the levels found in the tap water samples. Based on the concentrations of the sweetener, the researchers estimated that swimmers released more than 7 gallons of urine—enough to fill a medium-size trash bin—in a 110,000-gallon pool in one instance, and nearly 20 gallons in a 220,000-gallon pool (one-third the size of an Olympic-size pool) in another instance.
More information: Sweetened Swimming Pools and Hot Tubs, Environ. Sci. Technol. Lett., Article ASAP, pubs.acs.org/doi/abs/10.1021/acs.estlett.7b00043
Abstract
Nitrogenous organics in urine can react with chlorine in swimming pools to form volatile and irritating N-Cl-amines. A urinary marker is desirable for the control of pool water quality. The widespread consumption of acesulfame-K (ACE), a stable synthetic sweetener, and its complete excretion in urine, makes it an ideal urinary marker. Here we report the occurrence of ACE and its potential application in swimming pools and hot tubs. First, we developed a new method for achieving high-throughput analysis of ACE without preconcentration or large-volume injection. Analysis of more than 250 samples from 31 pools and tubs from two Canadian cities showed ACE in all samples. Concentrations ranged from 30 to 7110 ng/L, up to 570-fold greater than in the input tap water. The level of dissolved organic carbon was significantly greater in all pools and tubs than in the input water. Finally, we determined the levels of ACE over 3 weeks in two pools (110000 and 220000 U.S. gallons) and used the average ACE level to estimate the urine contribution as 30 and 75 L. This study clearly shows the human impact in pools and tubs. This work is useful for future studies of the human contribution to DBP formation, epidemiological assessment of exposure, and adverse health effects in recreational facilities.
Journal information: Environmental Science & Technology Letters
Provided by American Chemical Society