Tracking real-time proton induced radiation chemistry in water

Proton therapy is a promising form of radiation treatment used to kill cancerous cells and effectively halt their rapid reproduction. While this treatment can also be delivered in different modalities (i.e. electrons and X-rays), proton therapy limits damage to healthy tissue by depositing energy in a highly localized dose volume.
The fundamental understanding for proton therapy is contained in the radiation induced water chemistry that occurs immediately after the interaction. This is because as much as 66 percent of the radiation deposited into a tumor volume is initially absorbed by water molecules in the cancer cells. The ensuing processes are therefore a subject of considerable scientific interest.
"It is at those fundamental levels that the seeds for the subsequent radiation chemistry are sown," explained Brendan Dromey, the lead researcher on this project and a reader in the Centre for Plasma Physics at Queen's University Belfast. "And it's from there that we can begin to build models from first principles that will allow us to gain a full understanding of how these early processes ultimately affect tumor cell death."
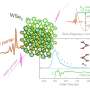
When energetic protons enter water, they can ionize the water molecules generating free electrons. In response, nearby water molecules can shift themselves so that their positive sides orient towards these freed electrons and shield their negative charge. To date, methodology to monitor the earliest stages of this process relied on "indirect scavenging techniques." This, however, requires the use of chemical additives that simultaneously increase the complexity of the observation. The new approach substitutes chemical scavengers for an experimental setup with enhanced temporal resolution.
Dromey and his colleagues in Sweden, Germany and Northern Ireland describe their work this week in the journal Applied Physics Letters.
"To use an analogy from photography, the existing methodology had a time resolution that functioned like a camera with slow shutter speed. If processes, such as these initial chemical changes, were progressing quickly, the slow shutter speed meant that one wouldn't capture the details of the movement and the image generated would be blurred. The new set-up and methodology that we outline in our article operates like a camera with a fast shutter speed. It allows us to capture rapid chemical evolution in high detail," Dromey said.
"Since our methodology does not use scavenging chemicals, we can work with water molecules in a pristine, controlled environment. Moreover, our technique features a fundamental time resolution that is less that one picosecond or one trillionth of a second. Even when we factor in diagnostics, the time resolution is less than five picoseconds. We can now track the radiation chemistry that follows the initial ionization of water molecules as they unfold in real-time," Dromey said, whose research is also funded by the Engineering and Physical Sciences Research Council in the U.K.
"Two major innovations account for this improvement. First, we use bursts of protons accelerated by the high power TARANIS laser facility in Queen's University Belfast. The sub-picosecond acceleration of an initially cold proton population permits the generation of an ultrafast pulse with low inherent thermal spread. Second, we use the same laser to generate both the pulse of protons and the probe that allows us to track the progress of the radiation chemistry. This eliminates the electrical jitter that one finds in more traditional, radiofrequency cavity based systems," Dromey said. "That said, it is important to note that in terms of energy stability and beam quality there is still significant development required for laser-based accelerators to match the performance of these machines."
Lovisa Senje, a doctoral student from the Department of Physics at Lund University and lead author on the paper, added, "The ultrashort proton pulses produced in our experimental set-up, in combination with the high number of protons per pulse, lead to a unique possibility of studying how water reacts to extreme irradiation by protons. We can actually see that under these conditions the processes following energy deposition of protons in water change."
"One of the most interesting things that we have discovered with the benefit of better temporal resolution is that there appears to be a delay in the formation of the absorption band of solvated electrons after exposure to protons," Dromey said. "This was surprising because past research suggests that you don't typically see this delay when you expose water molecules to X-rays or electrons. Our future work will focus on systematically exploring this delay further."
More information: L. Senje et al, Experimental investigation of picosecond dynamics following interactions between laser accelerated protons and water, Applied Physics Letters (2017). DOI: 10.1063/1.4977846
Journal information: Applied Physics Letters
Provided by American Institute of Physics