October 17, 2016 report
Using light to move electrons and protons
(Phys.org)—In some chemical reactions both electrons and protons move together. When they transfer, they can move concertedly or in separate steps. Light-induced reactions of this sort are particularly relevant to biological systems, such as Photosystem II where plants use photons from the sun to convert water into oxygen.
To better understand how light can lead to the transfer of protons in a chemical reaction, a group of researchers from the University of North Carolina, Shanxi University in China, and Memorial University in Newfoundland have conducted adsorption studies on a new family of experiments to observe the transition that occurs when protons transfer between hydrogen-bonded complexes in solution . They provide evidence for new optical transitions characteristic of the direct transfer of a proton. This report recently appeared in the Proceedings of the National Academy of Sciences.
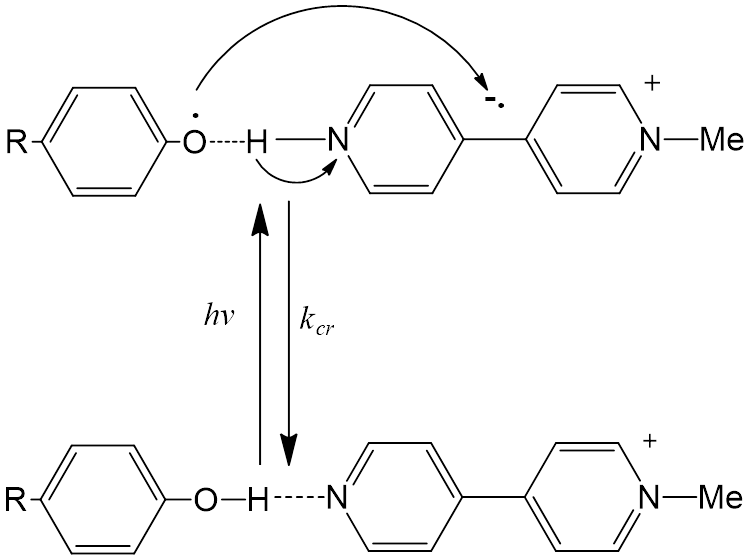
N-methyl-4,4'-bipyridinium cation (MQ+) serves as proton acceptor, where a proton will add to the non-methylated pyridinium amine. If proton transfer occurs, then MQ+ will form a radical cation (MQH+•) whose absorbance spectra in the UV/visible range can be compared to N, N'-dimethyl-4, 4'-bypyridinium (MV2+).
By using ultrafast laser flash photolysis measurements, they found direct evidence for a low energy absorption band between p-methoxyphenyl and the mehylviologen acceptor, MQ+. It appears at 360 nm and as early as 250 fs after the laser pulse. Based on these properties, it is clearly the product of proton transfer from the phenol to give MeOPhO•—H-MQ+.
The appearance of this reaction involving the transfer of both an electron and proton after absorbing a single photon is supported by the vibrational coherence of the radical cation and by it characteristic spectral properties. By inference, related transitions, which are often at low intensities, could play an important role in the degradation of certain biological molecules, such as DNA.
The appearance of these absorption bands could have theoretical significance. They demonstrate a way to use simple spectroscopic measurements to explore the intimate details of how these reactions occur in nature. This provides new physical insight into processes that could be of broad biological and chemical relevance.
More information: Christopher J. Gagliardi et al. Direct observation of light-driven, concerted electron–proton transfer, Proceedings of the National Academy of Sciences (2016). DOI: 10.1073/pnas.1611496113
Abstract
The phenols 4-methylphenol, 4-methoxyphenol, and N-acetyl-tyrosine form hydrogen-bonded adducts with N-methyl-4, 4′-bipyridinium cation (MQ+) in aqueous solution as evidenced by the appearance of low-energy, low-absorptivity features in UV-visible spectra. They are assigned to the known examples of optically induced, concerted electron–proton transfer, photoEPT. The results of ultrafast transient absorption measurements on the assembly MeOPhO-H—-MQ+ are consistent with concerted EPT by the instantaneous appearance of spectral features for MeOPhO·—-H-MQ+ in the transient spectra at the first observation time of 0.1 ps. The transient decays to MeOPhO-H—-MQ+ in 2.5 ps, accompanied by the appearance of oscillations in the decay traces with a period of ∼1 ps, consistent with a vibrational coherence and relaxation from a higher υ(N-H) vibrational level or levels on the timescale for back EPT.
Journal information: Proceedings of the National Academy of Sciences
© 2016 Phys.org