Sea urchin spines could fix bones
More than 2 million procedures every year take place around the world to heal bone fractures and defects from trauma or disease, making bone the second most commonly transplanted tissue after blood. To help improve the outcomes of these surgeries, scientists have developed a new grafting material from sea urchin spines. They report their degradable bone scaffold, which they tested in animals, in the journal ACS Applied Materials & Interfaces.
Physicians have various approaches at hand to treat bone defects: Replacement material can come from a patient's own body, donated tissue, or a synthetic or naturally derived product. All of these methods, however, have limitations. For example, current bioceramics, such as hydroxyapatite, that have been used as scaffolds for bone repair tend to be weak and brittle, which can lead to pieces breaking off. These pieces can then move into adjacent soft tissue, causing inflammation. Recent studies have shown that biological materials, such as sea urchin spines, have promise as bone scaffolds because of their porosity and strength. Xing Zhang, Zheng Guo, Yue Zhu and colleagues wanted to test this idea in more detail.
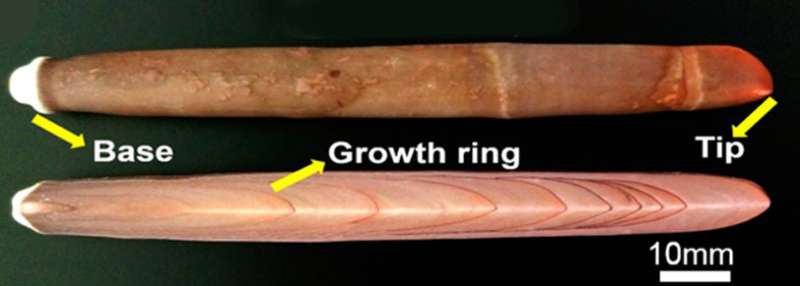
Using a hydrothermal reaction, the researchers converted sea urchin spines to biodegradable magnesium-substituted tricalcium phosphate scaffolds while maintaining the spines' original interconnected, porous structure. Unlike hydroxyapatite, the scaffolds made from sea urchin spines could be cut and drilled to a specified shape and size. Testing on rabbits and beagles showed that bone cells and nutrients could flow through the pores and promote bone formation. Also, the scaffold degraded easily as it was replaced by the new growth. The researchers say their findings could inspire the design of new lightweight materials for repairing bones.
More information: Lei Cao et al. Lightweight Open-Cell Scaffolds from Sea Urchin Spines with Superior Material Properties for Bone Defect Repair, ACS Applied Materials & Interfaces (2017). DOI: 10.1021/acsami.7b01645
Abstract
Sea urchin spines (Heterocentrotus mammillatus), with a hierarchical open-cell structure similar to that of human trabecular bone and superior mechanical property (compressive strength ∼43.4 MPa) suitable for machining to shape, were explored for potential applications of bone defect repair. Finite element analyses reveal that the compressive stress concentrates along the dense growth rings and dissipates through strut structures of the stereoms, indicating that the exquisite mesostructures play an important role in high strength-to-weight ratios. The fracture strength of magnesium-substituted tricalcium phosphate (β-TCMP) scaffolds produced by hydrothermal conversion of urchin spines is about 9.3 MPa, comparable to that of human trabecular bone. New bone forms along outer surfaces of β-TCMP scaffolds after implantation in rabbit femoral defects for one month and grows into the majority of the inner open-cell spaces postoperation in three months, showing tight interface between the scaffold and regenerative bone tissue. Fusion of beagle lumbar facet joints using a Ti-6Al-4V cage and β-TCMP scaffold can be completed within seven months with obvious biodegradation of the β-TCMP scaffold, which is nearly completely degraded and replaced by newly formed bone ten months after implantation. Thus, sea urchin spines suitable for machining to shape have advantages for production of biodegradable artificial grafts for bone defect repair.
Journal information: ACS Applied Materials and Interfaces
Provided by American Chemical Society