Scientists present the smallest member of the CRISPR-Cas9 family developed to date
Scientists at the Center for Genome Engineering, within the Institute for Basic Science (IBS), in collaboration with KIM Eunji (ToolGen Inc.) and KIM Jeong Hun (Seoul National University) have engineered the smallest CRISPR-Cas9 to date, delivered it to the muscle cells and in the eyes of mice via adeno-associated viruses (AAV) and used it to modify a gene causing blindness. Published on Nature Communications, this CRISPR-Cas9 system, originated from Campylobacter jejuni (CjCas9), is expected to become a useful therapeutic tool against common and "undruggable" disease targets.
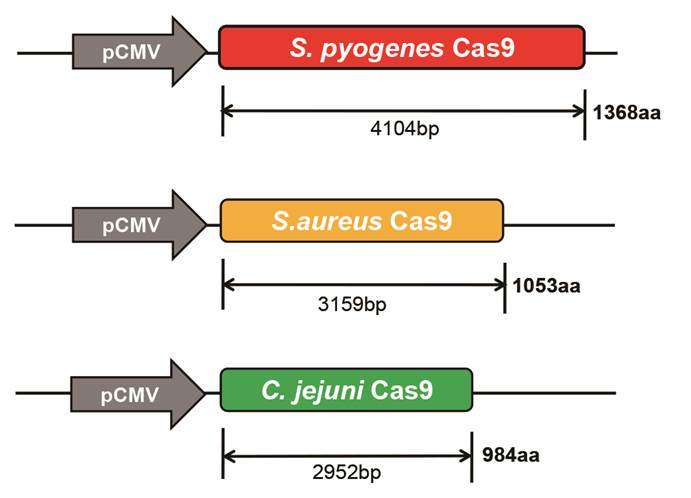
CRISPR-Cas9 is the buzzword amongst molecular biologists. It is an innovative, cheap and precise technique to edit genes. Cas9 is the "gene scissors" protein: It creates cuts on the target gene in precise locations indicated by the guide RNA. In order for CRISPR-Cas9 complex to reach the target DNA, it has to be delivered via plasmids or viruses. "AAV is an efficient and safe vector to express a gene of interest in vivo and has been used widely in gene therapy," explains KIM Jin-Soo, director of the IBS Center for Genome Engineering and corresponding author of the study. Naturally, Cas9 is used by several bacteria as an immunity weapon; it is needed to cut viral DNA which could damage the bacteria. The most common version of the CRISPR-Cas9 technique uses Cas9 derived from the bacterium Streptococcus pyogenes. However, this protein is made of 1,368 aminoacids and it is too large to be delivered and packaged in AAV. Even if scientists split it up into two parts, each packaged in a different virus, other issues arise: A double amount of viruses need to be delivered and the split Cas9 is less active than the intact SpCas9. Staphylococcus aureus Cas9 is also used for gene editing. It is slightly smaller (1,053 aminoacids), so that it can just fit inside the AAV, but does not leave enough space for other proteins.
In this study, the team found that CjCas9 is both efficient and small. It has 984 aminoacids and it can be packed in AAV together with more than one guide RNAs as well as with a fluorescent reporter protein.
In order to use a bacterial protein for gene editing, scientists had to optimize some aspects of the technique. They designed a short DNA sequence immediately following the DNA sequence targeted by the Cas9, called Protospacer Adjacent Motif (PAM). Each different Cas9 needs a specific PAM sequence, otherwise it will not be able to bind to and cleave the target DNA sequence. Secondly, they had to modify the length of the guide RNA.
Afterwards, IBS scientists packaged the new CRISPR-Cas9 complex into AAV, together with two guide RNAs and a fluorescent reporter protein, to mutate genes in mice's muscles and eyes. They concentrated on two genes involved in the age-related macular degeneration (AMD), one of the leading causes of blindness in adults. One gene is a common therapeutic target for ADM, called Vascular endothelial growth factor A (VEGF A), the other one is a transcription factor that activates the transcripction of VEGF A and it is known as HIF-1a. Unlike VEGF A, HIF-1a has not been considered as a drug target. So-called'undruggable'genes, such as transcription factors in general, cannot be targeted directly by antibodies and other biological or chemical drugs. In this study, the research team proved that CjCas9 delivered to the retina via AAV can inactivate Hif1a and VEGF A in mice efficiently and reduced the area of choroidal neovascularization (CNV).
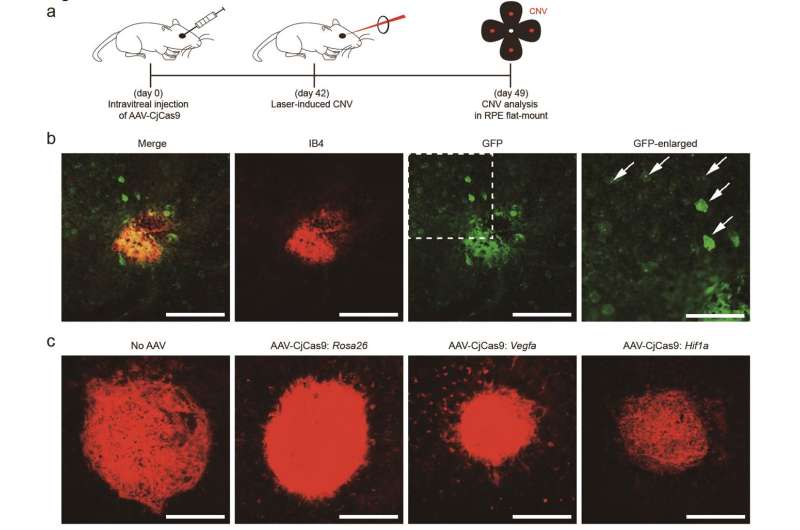
Intraocular injections of AAV-packaged CRISPR-CjCas9 could be beneficial to treat various retinal diseases and systemic diseases. "CjCas9 is highly specific and does not cause off-target mutations in the genome," explains KIM Jin-Soo.
Hif1a gene target sequences are the same in both mice and humans, thereby the method presented in this study could be used in the future for the treatment of ADM in human patients. By paving the way to the application of CjCas9 against 'undruggable' genes or non-coding sequences, this technology can broaden the range of therapeutic targets, making the entire human genome potentially druggable.
More information: Eunji Kim et al, In vivo genome editing with a small Cas9 orthologue derived from Campylobacter jejuni, Nature Communications (2017). DOI: 10.1038/ncomms14500
Journal information: Nature Communications
Provided by Institute for Basic Science