Real-time imaging of cell components including DNA
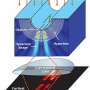
Optical microscopes that use lenses to bounce photons off objects have trouble distinguishing nanometer-scale objects smaller than the imaging beam's wavelength, such as proteins and DNA. An innovative 'hyperlens' designed at A*STAR can overcome optical diffraction limits by capturing high-resolution information held by short-lived or evanescent waves lurking near a target's surface.
Hyperlens devices—composed of thin stacks of alternate metal and plastic layers—have raised prospects for capturing living biological processes in action with high-speed optics. Key to their operation are oscillating electrons, known as surface plasmons, that resonate with and enhance evanescent waves that appear when photons strike a solid object. The narrow wavelengths of evanescent beams give nanoscale resolution to images when the hyperlens propagates the images to a standard microscope.
Mass-production of current hyperlenses has stalled however because of their intricate fabrication— up to 18 different layer depositions may be required, each with stringent requirements to avoid signal degradation. "For perfect imaging, these layers need precisely controlled thickness and purity," says Linda Wu, from the A*STAR Singapore Institute of Manufacturing Technology. "Otherwise, it's hard to magnify the object sufficiently for a conventional microscope to pick up."
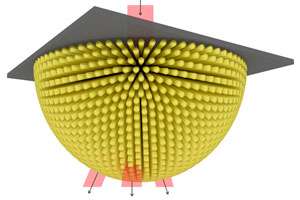
Wu and her co-workers proposed a different type of hyperlens that eliminates the need for multiple interfaces in the light propagation direction—a major source of energy loss and image distortion. The team's concept embeds a hemispherical array of nanorods into a central insulating core, giving the hyperlens a shape similar to a thorny sea urchin. This geometry enables more efficient harvesting of evanescent waves, as well as improved image projection.
"For the sea urchin geometry, the nano-sized metallic structures align in the same direction of the light propagation direction, and they are much smaller than the wavelength of applied infrared light," explains Wu. "Therefore the light doesn't 'see' any obstacles, and propagates effectively and naturally, without loss."
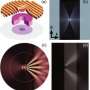
The researchers' simulations revealed the spiky hyperlens could separate the complex wave information into its component frequencies, and then transmit this data to the microscope as an intense, easy-to-spot band. This approach was also efficient – it proved capable of resolving intricate objects, 50 to 100 nanometers wide, without the need for image post-processing.
Wu notes that fabricating sea urchin hyperlenses should be much simpler than multi-layered structures. "The nano-sized metallic structures could be formed using pores and templates into flexible lenses, with no real size limitations," she says. "This hyperlens could be an important tool for real-time bio-molecular imaging."
More information: Ankit Bisht et al. Hyperlensing at NIR frequencies using a hemispherical metallic nanowire lens in a sea-urchin geometry, Nanoscale (2016). DOI: 10.1039/c5nr09135g
Journal information: Nanoscale