When ice and snow melt away into streams and groundwater, road salt goes with it

At the edge of Behrend Fields, where a footpath leads back to a one-acre parking lot, Pam Silver bends down and scoops a handful of snow into a small plastic cup.
It's February, 2016. Silver, distinguished professor of biology at Penn State Erie, The Behrend College, hands the cup to Mali Lubic, one of a dozen biology and environmental science students who have volunteered to collect snow from 110 locations on campus. Lubic fits it into a tote with a four-acre grid mapped on the lid. Fresh snow blows around her as she seals it.
"I could do without that wind," Lubic says.
They've been out here for an hour, cupping snow from the banks of Trout Run, and from the woods above it, and from a spot just beyond the track-and-field team's shot-put pad. They have samples from the bike trail, the third- and first-base edges of the softball field, and from the base of the directional sign that guides traffic off the four-lane, $180 million Bayfront Connector that runs along the north edge of campus.
Silver, an aquatic ecologist, is collecting the snow to get at the rock salt that has been mixed into it. She wants to know where the salt that is washed, plowed, and blown off campus paths and roads goes after the snow melts. In the lab, she, Lubic and other students will melt the samples and measure their electrical conductivity, an indication of how much salt is in each. That data will refine a map that shows elevated concentrations of salt not only on the college's parking lots and walkways, but also along Trout Run, which flows into Fourmile Creek, which empties into Lake Erie, the primary source of drinking water for the 280,000 residents of Erie County.
When it snows again, and the bank teller or the postal clerk or the cart boy at the supermarket, stomping through the winter slop, jokes that Erie's winters never really end, Silver will have data that proves it.
Four months later, on an 88-degree day, she will check the salinity in Glenhill Stream, a three-skip stone's throw from her office in the Benson Building. It will be nine times higher than it should be.
Benefits and costs
Silver accepts that some level of salt is necessary. The 4,300 students and 700 employees who live, study, and work at Penn State Behrend do so through the winter season, which in Erie typically produces more than 100 inches of snow.
"In the short term, the primary concern is safety," Silver explains. "We don't want people to be falling as they walk to class."

The campus Maintenance and Operations staff, which has provided data and logistical support for her study, applies more than 500 tons of rock salt to the college's walkways and parking lots each year. They mix it with stone grit, which adds friction. The grit also reduces the cost of the salt, which is purchased in bulk from Morton Salt, which extracts more than 1.3 million tons of rock salt every year from a vast deposit 2,000 feet beneath Lake Erie. Even then, the cost is significant: The college will spend more than $39,000 on rock salt this year.
Parking lot curbs keep some of the salt on the pavement, reducing salt levels in adjacent areas. Culverts draw more of it into storm sewers, which eventually empty into the lake. An elaborate drainage system beneath the Bayfront Connector directs runoff into a constructed wetland and away from nearby homes. A second wetland absorbs salt overspray from the road.
The Pennsylvania Department of Transportation is mindful of its salt use: It applies nearly 27,500 tons of salt every year to 779 miles of Erie County roads, including highways. In 2003, when the Connector was completed, the department asked Silver to study the new road's impact on nearby animals.
"We had these brand-new, virgin wetlands that had never had water in them," she says. "It was a perfect starting point for a research study."
Data loggers purchased by the Pennsylvania Department of Transportation measured the level of salt in and near one of those wetlands for three years. Silver analyzed the data, and sediment from the bottom of the wetlands, to assess the impact on non-biting midges, which live in marshy conditions.
"Midges are a good indicator species," Silver explains. "They're easy to cultivate, their biology is well understood, and they live in the mud. If salt is going to accumulate, it will be there, in the sediment. We can see how it affects them."
Two years after the road opened, there were significantly fewer midges in the wetland designed to receive the runoff than in other constructed wetlands that were kept free of salt.
Silver would like to know the impact elevated salt concentrations have on other strands of the food web, including algae, salamanders, frogs, and fish. "Salinization of fresh water can cause immense ecological harm," she says. "All of the natural services that fresh water provides, including photosynthesis and the processing of leaf material—the processes that are not on our radar, but that keep us alive—are directly affected by it."
There is an economic cost as well: Recreational fishing is a $1.3 billion business in Pennsylvania, according to the state's Fish and Boat Commission. That disappears if the habitat can no longer sustain fish.
For now, however, Silver is focused on the salt runoff across Penn State Behrend's 854-acre campus. "If we can put the pieces together here," she says, "we will have a far more complete picture of the impact this salt is having on our environment, including our drinking water. If we can develop new methods for containing it, or for using it more efficiently, and we can point to that success, other, larger entities, including municipalities, might follow suit."
Seeing the problem

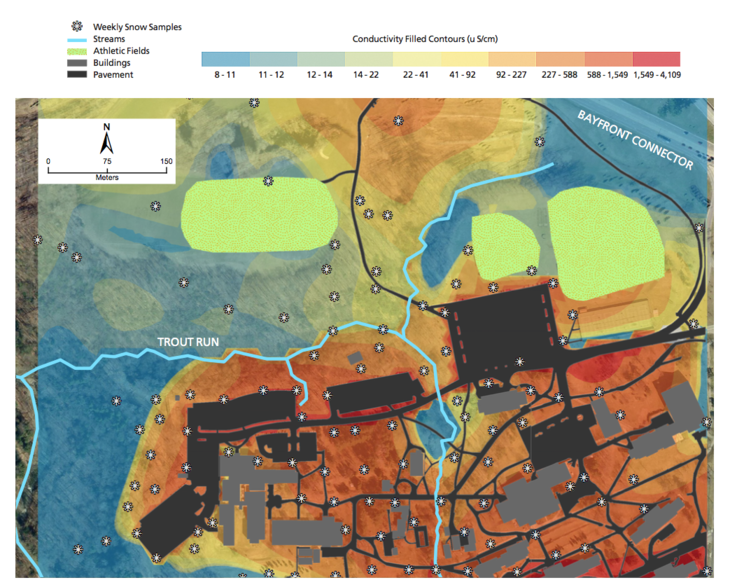
"To fix a problem, you have to first see it," says Michael Naber, a lecturer in geosciences. With help from Devin Beggs, a science major, Naber mapped the conductivity levels that Silver and her students recorded over a six-week period last winter. That data, drawn from the 110 sampling sites, allowed him to predict the presence of salt elsewhere on campus.
"People know this stuff's out there," he says. "They see it on their cars and their boots and on the carpets when they enter a building. What's eye-opening is how concentrated it is in some areas."
On his map, the small parking lot near Glenhill Farmhouse is alarm-button red. Some snow samples collected there had conductivity rates of more than 44,000 microsiemens/cm, which is 40 times the safe level for drinking water. At that concentration, the salt kills grass, algae, midges, and fish.
Over the next year, Naber will work with Michael Rutter, associate professor of statistics, to refine the salt-distribution projections on the map. They will use additional data, including samples from the southern edge of campus, where construction of a 250-bed residence hall is expected to begin in late 2017, altering the route of runoff to lower elevations.
In November, Silver will collect a trove of new information from three data loggers that she has placed in streams on campus. The loggers measure the water's conductivity every 15 minutes. By matching that data to weather reports, Silver should be able to determine how quickly conductivity spikes during storms and warming periods—and how long the salt concentrations remain elevated.
Additional samplers will be secured to the grates on 15 campus storm drains. That effort is being coordinated by Tony Foyle, associate professor of geology, whose students will measure the conductivity of water that enters the drains during peak flow events, including storms. A spike in those readings would be particularly troubling: Medallions on the tops of the grates explain that the pipes beneath them drain into Trout Run.
Informed choices
The third phase of the project will involve faculty members with expertise beyond the biology and geology departments. Silver has a particular skill for opening her work to additional collaborators, including undergraduates.
"I talk a lot about this project," she says. "I keep putting it out there, saying, basically, 'This is what we're doing. Do you want to play?' People pick up the pieces of it that interest them."
High-school students from Northwest Pennsylvania Collegiate Academy are helping Luciana Aronne, a lecturer in chemistry, test Silver's snow samples for chloride.
Deborah Aruguete, assistant professor of environmental science, will provide additional analysis. She studies the impact on soils when toxic metals are released in automobile emissions. Those metals do even more damage when they are mixed with salt.
Silver has met with Joshua Shaw, associate professor of philosophy, to discuss the ethical ramifications of salt use. She also sees opportunities to work with colleagues in the college's communication and political science departments.
"Environmental problems don't begin with science," she explains. "They result from people needing or wanting something else. They are political, social, and economic problems, and to address them you really do need to get buy-in from the wider community. If we can put all these pieces together and present a clear picture of the impact this salt is having on our environment, people will say, 'Wow, I had no idea.' And then, maybe, they'll change their habits."
Silver and her students have talked through a long list of potential fixes, including higher curb heights and the use of salt brines—liquid salt mixtures—to treat roadways before snow falls. It takes four times more salt to remove ice after it forms than to prevent it before or during a snow event, a 2014 study by the New York State Department of Transportation found.
Other options include canopies, solar panels and enclosed paths that connect some buildings on campus. The Otto Behrend and Hammermill science buildings were connected in 2015, allowing students to move through the six-building School of Science complex without having to go outside.
That's an expensive fix. Silver's own approach—to blaze her own path, whenever possible, avoiding surfaces that have frozen or crusted over—is no more realistic: Most people prefer to take the clearest, most convenient route.
There she goes, though, with her tote full of samples, stepping off the cleared path and into deep snow. It grips her, like movie quicksand, clamping onto the full length of her legs. She trudges forward, Lubic a few steps behind, stirring up fine clouds of light, white snow. The path, and the salt that cleared it, is just one piece of a much larger puzzle, a reminder that every convenience comes with some consequence.
Provided by Pennsylvania State University