Titanium phosphate displays unusual transformations at high pressure
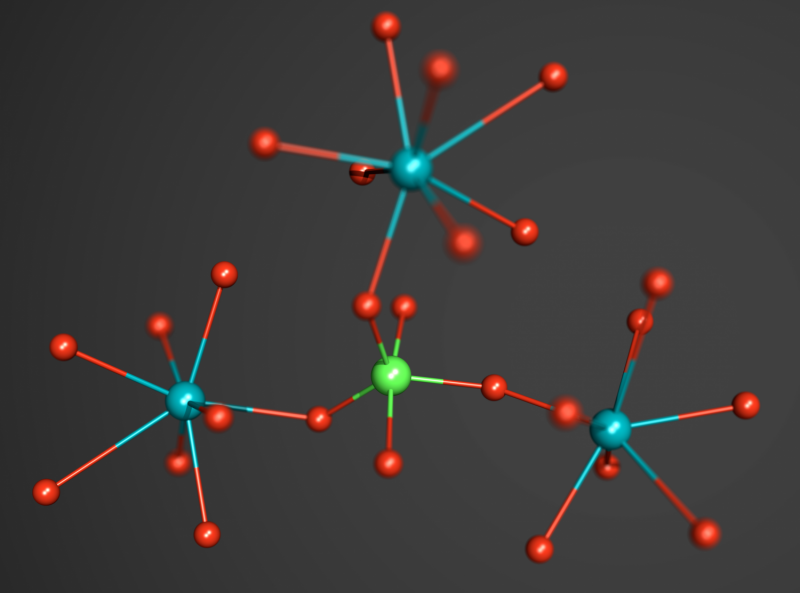
Scientists at DESY and the European Synchrotron Radiation Facility ESRF have discovered a new and very unusual form of phosphorus compound. In high-pressure experiments, a team led by Dr. Maxim Bykov from the University of Bayreuth found evidence for a configuration where a central phosphorus atom is bound to five surrounding oxygen atoms (PO5). Such a configuration is very rare and it is the first time that PO5 groups were found in an inorganic phosphate compound. The researchers report their findings in the scientific journal Angewandte Chemie International Edition.
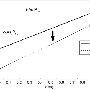
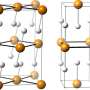
The scientists studied titanium orthophosphate (TiPO4) at pressures of up to 56 Giga-Pascal (GPa), more than 500 000 times the average pressure at sea level. To achieve this pressure, they used a device called a diamond anvil cell, where the sample is compressed between two tiny diamond anvils. With the intense X-ray beam at DESY's synchrotron radiation source PETRA III and using a novel methodology for single crystal diffraction experiments at high pressures they were able to distinguish and characterize four phase transitions that appeared during the compression of titanium orthophosphate to over half a million atmospheres. "In the last phase discovered at the highest achievable pressure, a central phosphorus atom was bound to – or coordinated by – five surrounding oxygen atoms," explains Bykov.
Phosphorus plays a key role in the chemistry of life and many technological applications. Unusually high coordination numbers of cations like phosphorus are often associated with interesting physical properties like high density, ultra-high hardness or low compressibility, which are very relevant for engineering applications. Pentacoordinated (five-coordinated) phosphorus is expected to be especially chemically active and thus may play a significant role in the synthesis of novel organic or metal-organic compounds. The experimental demonstration that phosphorus could form PO5 polyhedra suggests that similar compounds may appear as intermedia (metastable, short-time leaving, and hardly or not at all detectable) products of different chemical processes involving phosphorus and oxygen. "The creation of new compounds based on PO5 groups could have significant implications for industrial applications if someone finds a way to recover these materials at ambient conditions and study their physical properties," says Bykov.
When they started their experiment, Bykov and his colleagues were actually looking to examine another phenomenon: titanium orthophosphate shows very unusual magnetic properties. Under normal pressure it forms a so called quasi-one-dimensional antiferromagnetic system. When it is cooled down, TiPO4 loses this form of magnetism and transforms to a diamagnetic state. This process is called spin-Peierls transition. The scientists were now able to prove that this transition also happens when TiPO4 is put under high pressure (above 7 GPa). "The result has important consequences for studying this process in more detail. It both improves our understanding of the spin-Peierls transitions and reveals new possibilities to examine this transition without low temperatures" explains Bykov.
More information: Maxim Bykov et al. High-Pressure Phase Transformations in TiPO4: A Route to Pentacoordinated Phosphorus, Angewandte Chemie International Edition (2016). DOI: 10.1002/anie.201608530
Journal information: Angewandte Chemie International Edition
Provided by Helmholtz Association of German Research Centres