November 14, 2016 report
'Super-dry' reforming reaction converts greenhouse gases to useful intermediates
(Phys.org)—A new "super-dry" carbon dioxide reforming reaction consumes two waste products, carbon dioxide and methane, and produces gases that can be used to make synthetic fuels and other important products.
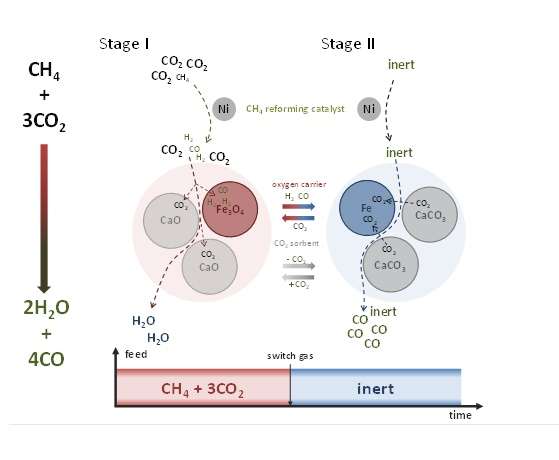
Researchers from Ghent University in Belgium, led by Dr. Vladimir Galvita have developed a nickel-catalyzed carbon reforming reaction scheme that involves the use of calcium oxide as a carbon dioxide sorbent and iron oxide as a solid oxygen carrier. This process does not involve temperature swings, allowing for better carbon monoxide production, and their two-flow system eliminates unwanted back reactions. Their work appears in a recent issue of Science.
In an effort to decrease CO2 production, scientists have developed methods to convert CO2 to helpful starting materials that can be used to produce synthetic energy sources. These methods involve reducing CO2. The most commercially feasible method is a process called dry reforming of methane, which produces syngases, CO and H2. This reaction needs to be a "dry" reaction because in the presence of water, the more energetically favored water gas shift reaction occurs. In this reaction carbon monoxide reacts with water to re-form carbon dioxide. Eliminating water from these reactions has proved to be an active area of research.
In the current study, Buelens et al. used calcium oxide as a CO2 sorbent in which calcium carbonate is formed. This has several benefits that that has allowed a higher carbon monoxide yield and an opportunity to remove water that is formed from the oxidation of methane.
First, from an economic and practical standpoint, because CO2 is removed in situ, the feed gas can be of lower stock quality. Secondly, the formation of calcium carbonate can be coupled with methane reformation and iron oxide reduction resulting in a more energetically favorable process. Then, when calcium carbonate decomposes into CO2 and CaO, the carbon dioxide is reduced to CO over the iron oxide oxygen carrier. According to the authors, it is at this point that the feed is switched to inert gas to regenerate the system.
They obtained a 45% higher CO yield, but this yield could be even higher by optimizing conditions. The higher efficiency of this reaction is due in large part by employing Le Chatlier's Principle.
Importantly, their two-flow reaction set-up seems to have some versatility that prior dry reforming reactions lacked either by changing the gas feedstock ratios or by changing to a multi-reactor configuration.
The applications of this technique, according to lead author, Lukas Buelens is that "with this process, we intensify the conversion of CO2 by making maximal use of CH4 as reducing gas. The generated CO can be used directly or combined with a green H2 source for the production of chemicals or fuels."
Additionally, their initial flow system uses a less expensive nickel catalyst because carbon deposition has been eliminated. Their system is more efficient for CO2 utilization than prior dry reforming reactions and may serve as a model for optimized CO2 conversion.
More information: L. C. Buelens et al. Super-dry reforming of methane intensifies CO2 utilization via Le Chateliers principle, Science (2016). DOI: 10.1126/science.aah7161
Abstract
Efficient CO2 transformation from a waste product to carbon source for chemicals and fuels will require reaction conditions that effect its reduction. We develop a "super-dry" CH4 reforming reaction for enhanced CO production from CH4 and CO2. We used Ni/MgAl2O4 as a CH4 reforming catalyst, Fe2O3/MgAl2O4 as a solid oxygen carrier, and CaO/Al2O3 as a CO2 sorbent. The isothermal coupling of these three different processes resulted in higher CO production compared with conventional dry reforming by avoiding back reactions with water. The reduction of iron oxide was intensified by CH4 conversion to syngas over Ni and by CO2 extraction and storage as CaCO3. CO2 is then used for iron reoxidation and CO production exploiting equilibrium shifts effected with inert gas sweeping (Le Chatelier's Principle). Super-dry reforming utilizes up to three CO2 molecules per CH4 and offers a high CO space-time yield of 7.5 mmol CO per second per kilogram of iron at at 1023 Kelvin.
Journal information: Science
© 2016 Phys.org