Molecules change shape when wet
In two recent publications in the Journal of Chemical Physics and in the Journal of Physical Chemistry Letters, researchers led by Melanie Schnell from the Max Planck Institute for the Structure and Dynamics of Matter at CFEL show that water promotes the reshaping of crown ethers and biphenyl molecules, two classes of chemically fascinating molecules. Crown ethers are key systems in catalysis, separation and encapsulation processes, while biphenyl-based systems are employed in asymmetric synthesis and drug design.
Water has profound implications in our world. From its known, yet not fully understood role in mediating protein folding dynamics and proton transport in membranes, water is a key player, influencing the mechanics of many biological and synthetic processes. In the present studies, the researchers used high-resolution rotational spectroscopy to investigate the structural effects that water induces in two types of molecular systems with different roles in the chemical realm. Isolated micro-solvated molecules in the gas phase have become an appealing target to reveal the stepwise hydration of molecular systems. The Hamburg group of Melanie Schnell took this route to reveal the effects that organic molecules undergo when the first water molecules bind around them, forming the so-called first solvation shell.
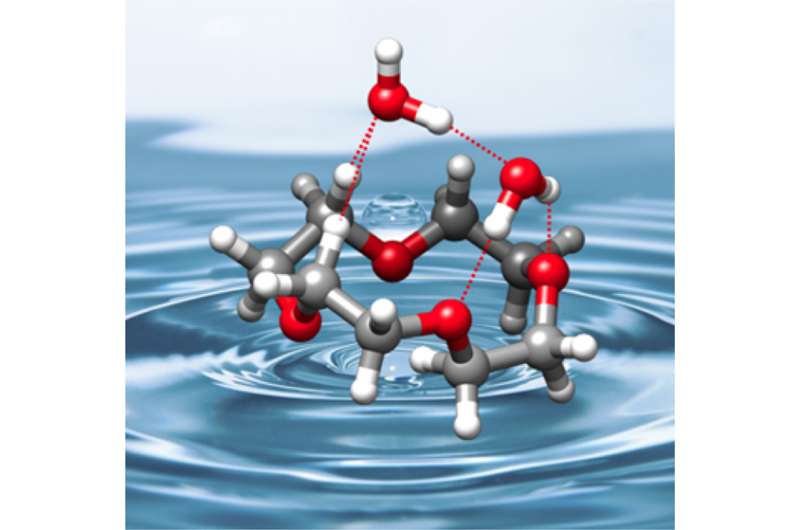
Crown ethers are cyclic molecules in a motif that resembles a crown. They have an extraordinary affinity for catching cations inside the crown. This function is either useful or harmful depending on the size of the crown and its consequent ability to bind smaller or larger cations such as potassium, sodium or lithium. Crown ethers are thus highly functional systems. In this work, the authors discovered that when binding to water, the crown ether's preferred shape changes. "The unexpected structural changes induced by the hydration of the crown ethers reveals new roads for host-guest interactions to take place," says Cristóbal Pérez, postdoc at the MPSD and first author of this work. The expected efficiency for catching other species may be altered in the presence of water. Given the abundance of water at the molecular scale where many biological processes take place, this revelation is important for chemists working with catalysis where crown ethers are employed.
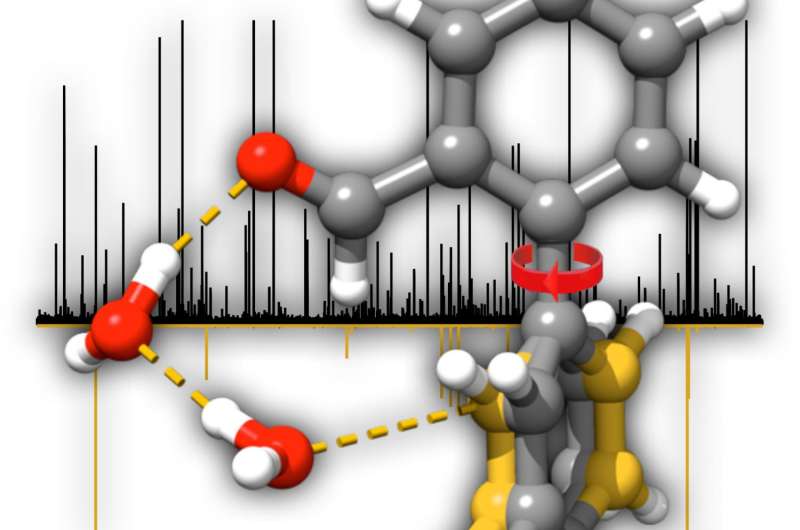
Biphenyl-based systems have a core consisting of two benzene (C6H6) rings connected via an axis. By overcoming a small energy barrier, the two rings are allowed to rotate with respect to each other. Clockwise and counterclockwise rotation generates mirror images of the same molecule that are not superimposed onto each other and can thus be termed chiral. Identifying and assigning the mirror image forms of chiral molecules are key steps in drug design in the pharmaceutical industry. As an example, the biphenyl motif occurs in drug designs against tuberculosis. In this study, the researchers discovered that when hydrated, the biphenyl system uses two water molecules to form what the authors call a "water-wire." The water wire links the two rings of the biphenyl motif and consequently locks the position of the two rings with respect to each other. With this locking mechanism due to the presence of water, a measurable change in the angle between the rings is observed. "The observed phenomenon provides us with new clues to how water may mediate the interactions between a molecule and its potential receptor," says Sérgio Domingos, postdoc at the MPSD and first author of this work. The observed structural changes induced by water are insightful on the role of hydration for the regulation of more complex biological processes taking place where water is the dominant medium.
More information: Cristóbal Pérez et al. Water-Induced Structural Changes in Crown Ethers from Broadband Rotational Spectroscopy, The Journal of Physical Chemistry Letters (2016). DOI: 10.1021/acs.jpclett.6b01939
Sérgio R. Domingos et al. Communication: Structural locking mediated by a water wire: A high-resolution rotational spectroscopy study on hydrated forms of a chiral biphenyl derivative, The Journal of Chemical Physics (2016). DOI: 10.1063/1.4966584
Journal information: Journal of Physical Chemistry Letters , Journal of Chemical Physics