Tailored chemistry links nanoparticles in stable monolayers

Just like carbon atoms in sheets of graphene, nanoparticles can form stable layers with minimal thicknesses of the diameter of a single nanoparticle. A novel method of linking nanoparticles into such extremally thin films has been developed at the Institute of Physical Chemistry of the Polish Academy of Sciences in Warsaw.
The chemical tailor cuts his coat according to... his nanoparticles
The tailoring successes to date of researchers synthesizing layers of nanoparticles would not be adequate to stage even the most modest of chemical fashion shows. Nanoparticles could be organized into single-particle layer thicknesses—that is, monolayers—but these structures were not stable because it was not possible to link nanoparticles together in a stable manner in monolayers until now.
"In recent years, our group at the Institute of Physical Chemistry of the Polish Academy of Sciences in Warsaw has been working on developing a universal platform for the synthesis of stable monolayers of nanoparticles. Today, we have proof that our 'tailored' method of chemically bonding nanoparticles in monolayers actually works," says Dr. Marcin Fialkowski, professor at IPC PAS, and demonstrates a tiny, layer, deposited on a plate, with the smallest possible thickness—equal to the diameter of a single nanoparticle of gold.
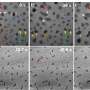
Monolayers of chemically stitched gold nanoparticles produced at the IPC PAS have surface areas of the order of square millimetres, and for obvious reasons, they are very delicate. Mechanically, they resemble acrylic plates—when subjected to forces, they initially deform elastically, after which they suddenly crack.
"Our monolayers are not large, because we only wanted to demonstrate the correctness of the concept of their synthesis. Nothing stands in the way of producing monolayers in the way that we propose with areas having many square centimetres," says Prof. Fialkowski.
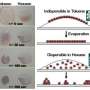
Nanoparticle layers have been produced for years at the interface between two immiscible liquids. When introduced into a heavier liquid, upon mechanical agitation, appropriately prepared nanoparticles flow out of it and distribute themselves randomly on the border with the lighter liquid. Order can established by compressing the nanoparticles with pistons from the side and thereby compacting them. Monolayers produced in this manner were hitherto not durable and when trying to remove them from the interface they simply fell apart. In turn, structures bound chemically, capable of surviving separation from the interface, always turned out to be either multi-layers or amorphous composites of nanoparticles.
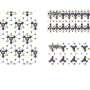
"Our monolayers are stable because we have linked the nanoparticles with special 'staples,' or linker molecules. Each linker joins together two adjacent nanoparticles by strong covalent bonds—that is, chemically", explains Dr. Tomasz Andryszewski (IPC PAS), lead author of the publication in the journal Chemistry of Materials.
The gold nanoparticles used in experiments at the IPC PAS have diameters of about five nanometres (billionths of a metre); the length of the linkers used is only one and a half. For such a short linker to bind together adjacent nanoparticles, these have to be appropriately shifted towards each other.
"The main difficulty in our work lay in the fact that we had to reconcile two requirements that were in principle opposite. Due to the length of the linker, we knew that the nanoparticles should be brought together to be a small distance apart, meaning that they would have to be subjected to relatively large forces. Therefore, we didn't want the nanoparticles to pop out of the interface. At the same time, we had to somehow prevent the nanoparticles from sticking together into random structures," says Dr. Andryszewski.
To meet these conditions, the nanoparticles were coated with small, specially designed molecules (ligands), which on one side contained amine groups (with nitrogen and hydrogen), and on the other, thiol groups (with sulphur and hydrogen). The thiol parts combined with the gold, whilst the amino parts located themselves on the outside of the nanoparticles and gave them a positive electric charge.
"The modified gold nanoparticles act as buoys with a large displacement. They locate themselves at the boundary between the liquids so durably that even strong agitation is not able to push them out. At the same time, they repel each other electrostatically. As a result, each nanoparticle is guaranteed a 'private space' around itself, necessary for the preservation of order," explains Ph.D. student Michalina Iwan (IPC PAS).
When the appropriately prepared nanoparticles had already been squeezed into monolayers at the interface, a linking substance was injected into the system. The cross-linking reaction, reminiscent of automatic stapling, took place at room temperature and at normal pressure, without the need for any initiators or catalysts. After the chemical anastomosis, the monolayer could be removed from the interface between the liquids, dried out, and even subjected to the action of strong solvents.
The physical properties of monolayers derived using tailored chemistry can be modified by selecting appropriate linkers. Longer, polymer linkers would allow the formation of monolayers with a higher elasticity. Using current-conducting linkers, it would in turn be possible to produce monolayers with specifically determined optoelectronic properties. The use of still other linkers could result in monolayers exhibiting a piezoresistive effect, i.e. changing their electrical conductivity under the influence of mechanical deformations. The new method of synthesis is also important for basic research—in the future, it will enable the direct investigation of things like the mechanical properties of single nanoparticles.
More information: Tomasz Andryszewski et al, Synthesis of a Free-Standing Monolayer of Covalently Bonded Gold Nanoparticles, Chemistry of Materials (2016). DOI: 10.1021/acs.chemmater.6b00922
Journal information: Chemistry of Materials
Provided by Polish Academy of Sciences