Drivers of evolution hidden in plain sight

Research led by the European Bioinformatics Institute (EMBL-EBI) and the University of Washington has shown that the biological diversity needed for evolution can be generated by changes in protein modifications. The findings, published today in Science, provide valuable insights into how different species adapt to different environments and could shed light on how pathogens evolve and become resistant to drugs.
"This study is about understanding how evolution works, which tells you how species adapt to changing environments over many generations," says Pedro Beltrao, a research group leader at EMBL-EBI. "For example, when you compare humans and chimps, they are obviously different, even though a good part of their genetic makeup is more or less the same. Our task is to figure out how diversity is generated, so that we can see in detail how life evolves. That helps us understand how plants and animals adapt and change, and how cancers or bacteria find their way around drugs."
A question of expression
Research into the drivers of genetic diversity has largely focused on gene expression, which controls how much of a given protein will be made, when, and in what tissue. However, the researchers found that a well-known cellular mechanism – one that controls how proteins acquire new functions – also plays a major role.
Proteins are controlled by other proteins by way of 'post-translational modification' (PTM). One type of PTM is phosphorylation: a rapid, versatile protein-regulation mechanism. During evolution PTMs can be acquired via mutations, which allows proteins to gain new functions, turn on or off at different times, and go to different places in the cell.
Previous studies comparing proteins in related species have shown very few mutations, so PTMs have not been considered to be a major factor in generating diversity. In today's study, the group found that only a few mutations are actually required to change these protein-modification sites. In other words, a small number of changes can have a big impact on how proteins and cells work.
"These mutations were hidden in plain sight – we could see them all along, but didn't know they could have such significant consequences," says Beltrao. "We only see it now after many years of developing and refining new experimental methods."
Change is the constant
Using experimental and computational methods, the researchers reconstructed the evolutionary history of phosphorylation sites – the modifications that can control proteins – in 18 different single-celled species. They determined how long these control points have existed, when they were acquired and how quickly they have changed across species over millions of years.
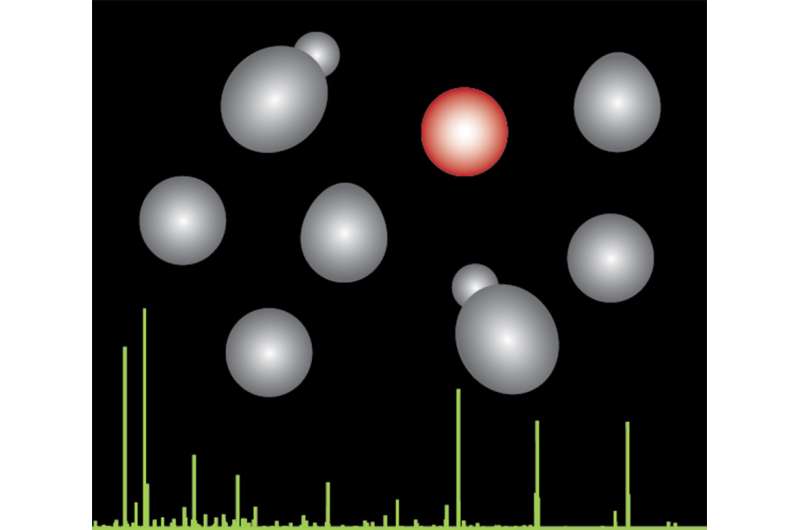
The group found that most of the phosphorylation sites had come about relatively recently in evolution, indicating that they are part of what make the species different – and a major contributor to evolutionary diversity.
"If a species needs to adapt to a new setting, it needs to generate a lot of diversity over many generations so that evolution has a pool of options to select from. One way for that to happen is through changes in gene expression, but changes in phosphorylation are equally effective," explains Beltrao.
Cancer: the expert lock picker
The diversity generated by PTMs is an important consideration in tackling cancer. Some cancer drugs stop tumours by blocking the signalling pathway that allows the tumour to grow, effectively placing a lock on a protein 'door'. But, through mutations, cancers find ways to create new PTMs and signalling events, effectively manufacturing millions of different keys. Most of the keys will be useless, but one is bound to fit the lock eventually, and the tumour can start growing again.
"Learning more about the role of PTMs in evolution also presents a much more reliable picture of how signalling proteins integrate and relay information inside the cell," adds Beltrao. "This in turn could present exciting new avenues for therapeutic research."
More information: Studer RA, et al., (2016) Evolution of protein phosphorylation across 18 fungal species. Science (in press); published online 14 October. DOI: 10.1126/science.aaf2144
Journal information: Science
Provided by European Bioinformatics Institute