Vital interplay between microorganisms and extracellular minerals
Some minerals abundant in soils and in aquatic and subsurface sediments electronically support microbial growth by supplying electrons or storing them as "environmental batteries," according to this new review article. Microbial cells derive chemical energy from metals associated with minerals outside the cell wall, but in many cases the microbial cell envelope is physically impermeable to minerals, or is not electrically conductive. Because of these barriers some microorganisms have evolved strategies to exchange electrons with extracellular minerals. The article outlines advances in understanding the mechanisms that allow needed electron exchange. Some mechanisms involve redox and structural proteins that form extensive electron transfer pathways. Others rely on microbial nanowires, which are conductive bacterial appendages anchored in the cell envelope, or they rely on ridged "cable bacteria."
Microorganisms with extracellular electron transfer capability have potential for environmentally sustainable biotechnological applications. Some bioremediation technologies already use such microbes to eat or degrade pollutants in soils, sediments, or groundwater. Biomining uses microbes to replace extractive strategies like extreme heat or toxic chemicals to leach desired elements from low-grade ores in a less polluting and more energy-efficient way. Microorganisms adept at mineral electron transfer have potential applications in catalysis, semiconductor manufacture, low-power microbial fuel cells, cancer treatment, and the production of clean biofuels and clever nanomaterials.
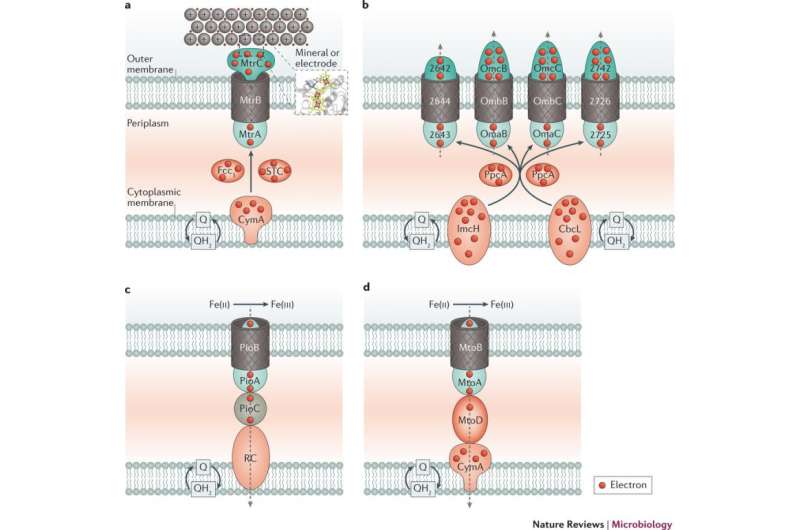
The cell envelopes of microorganisms have a cytoplasmic membrane that is the cell's primary barrier to the external environment. This membrane is also the nexus of the electron transfer essential for most microbial energy generation, but it often contains external structural elements that make it electronically nonconductive and physically impermeable to minerals. Microbial extracellular electron transfer overcomes these barriers by using a set of evolved specialized mechanisms for the exchange of electrons. These same strategies suggest that microorganisms with extracellular electron transfer capabilities have potential for biotechnological applications.
Biotechnologies already in place include immobilizing uranium at contaminated sites and recovering gold, copper, and other metals from low-grade ores. This review article summarizes the substantial progress made in the last decade in understanding the mechanisms of microbial extracellular electron transfer.
The authors summarize the advances, which include: the molecular identification and functional characterization of representative electron transfer pathways in four model microorganisms; the discovery of electron transfer reactions that occur over centimeters, a vast distance in the microbial universe; the suggestion that electron transfer pathways can be bidirectional; the direct transfer of electrons between microbial cells using nanowires or multicellular "cable bacteria" - even between cells of different species; minerals that act as electrical conductors or batteries; and some biotechnological applications that microbial electron transfer mechanisms can inform or inspire.
The authors also discuss knowledge gaps, including understanding inward electron transfer mechanisms; how to capture the molecular structure of active proteins at an atomic resolution; and how nanowires and cable bacteria actually work.
More information: Liang Shi et al. Extracellular electron transfer mechanisms between microorganisms and minerals, Nature Reviews Microbiology (2016). DOI: 10.1038/nrmicro.2016.93
Journal information: Nature Reviews Microbiology
Provided by Pacific Northwest National Laboratory