The role played by solvents at extreme pressure
Some living organisms thrive under extraordinary conditions, such as the deep sea. But just how do they withstand the enormous pressure?
Researchers at the Ruhr-Universität Bochum and the Technische Universität Dortmund have examined how the interactions between solvents and biomolecules change at high pressure. With infrared spectroscopy and computer simulations, they analysed the behaviour of the small molecule TMAO—short for trimethylamine oxide—in a pressure range from one bar to ten kilobars. These results could help us understand how organisms have adapted to life in the deep sea on the molecular level.
The team of the Bochum Chair for Theoretical Chemistry, headed by Prof. Dr. Dominik Marx, cooperated with the Dortmund Work Groups for Physical Chemistry of Prof. Dr. Roland Winter and Theoretical Physical Chemistry of Prof. Dr. Stefan M. Kast in the context of the Universitätsallianz Ruhr. They report their results in the journal Angewandte Chemie International Edition.
Life under extreme conditions
Not only micro-organisms but also larger animals, such as fish, thrive at extreme pressures in the deep sea. How they master this challenge on the molecular level has been little understood until now. It is known that, at high pressures, small molecules that stabilize protein structures accumulate in the cells of these creatures. One such molecule is TMAO. Just how it works, however, has remained a matter of speculation until now.
Spectra change systematically at rising pressure
The team of Roland Winter used Fourier transform infrared spectroscopy to record how the spectra of TMAO change with increasing pressure. In a diamond anvil cell constructed especially for the purpose, the researchers varied the pressure from normal conditions up to the kilobar range.
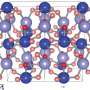
The vibration spectra obtained in this way provide insight into the structure of the molecule itself and also into the way in which its immediate solvent environment adapts to the pressure. The scientists observed that some bands of the spectrum shifted to higher frequencies, but also that individual peaks changed their form in a characteristic manner.
The two groups of Dominik Marx and Stefan Kast, both working theoretically, confirmed these findings with ab initio simulations and calculations based on liquid state theory. From the data, the team elucidated the molecular cause of the experimentally observed spectral changes.
More hydrogen bonds
At low pressure, the negatively polarized oxygen atom of the TMAO molecule creates three hydrogen bonds with the surrounding water molecules in almost all cases. At high pressure, however, according to the computer simulations, about half of all TMAO molecules have four hydrogen bonds. "That is a drastic change in the solvation behaviour. We have never seen anything like this before", says Dominik Marx.
"We can still only speculate whether this effect can actually explain how folded proteins and biomolecular processes in water together with TMAO can withstand extremely high pressures", adds Roland Winter. "This is exactly what we want to find out together in further studies in our High Pressure Research Group".
With their combination of methods, the Dortmund and Bochum researchers have the right tools at their disposal. "Coupling infrared spectroscopy with our theoretical methods based on the electronic structure of the molecules provides an outstanding tool for gaining new insights into the world of extreme biophysics, which is very difficult to access experimentally", says Stefan Kast.
More information: Sho Imoto et al, Toward Extreme Biophysics: Deciphering the Infrared Response of Biomolecular Solutions at High Pressures, Angewandte Chemie International Edition (2016). DOI: 10.1002/anie.201602757
Journal information: Angewandte Chemie International Edition
Provided by Ruhr-Universitaet-Bochum