Programmable materials find strength in molecular repetition
Synthetic proteins based on those found in a variety of squid species' ring teeth may lead the way to self-healing polymers carefully constructed for specific toughness and stretchability that might have applications in textiles, cosmetics and medicine, according to Penn State researchers.
"We looked at what is common among squid teeth proteins for all species of squid we studied," said Abdon Pena-Francesch, graduate student in engineering science and mechanics. "We observed which properties changed dramatically for each set of proteins."
Huihun Jung, a Ph.D. student in Melik Demirel's lab group, looked at four squid species from around the world—Hawaiian bobtail squid, long-finned squid, European squid and Japanese flying squid.
"It was a mystery why nature uses more than one protein to make the ring teeth in the suckers," said Demirel, professor of engineering science and mechanics. "Why did we need so many? It turns out that each has different mechanical properties."
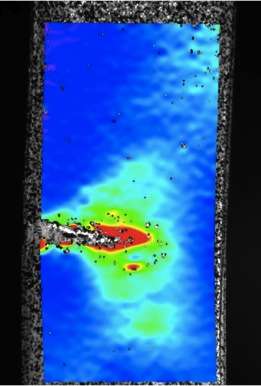
The proteins in ring teeth are semicrystalline, a combination of crystalline and amorphous pieces. The natural proteins also have varying repeats, amino acid strings that repeat themselves once or many times. These repeats alter the lengths of the protein. However, a clear understanding of the function of these repeats was not known.
After sequencing the various squid proteins, the researchers put together a variety of synthetic ring teeth proteins with varying numbers of repeats. They report their results in the current online issue of the Proceedings of the National Academy of Sciences.
"There has been a lot of work done making structures to mimic proteins," said Demirel, who is also a member of the Huck Institutes of the Life Sciences. "People have looked at the structure of proteins in silk, the elastin in skin, the resilin in insect wings and collagen in a large set of structures, but no one has looked at squid in this way. Squid mimics have not been done before."
Together with Benjamin Allen, research associate in biochemistry and molecular biology and the Huck Institutes, the Demirel group made varying lengths of amino acid strings—polypeptides—and found that in the synthetic material, toughness and extensibility increase as the molecular weight increases. The longer the polypeptide chain, the greater the molecular weight. They also found that the balance between elasticity—how much the material will stretch without deforming—and plasticity—the point at which it will deform—remained unchanged.
"We can control which amino acids we use, so we can control the molecular weights," said Pena-Francesch. "We can design each segment and see what fundamentals of mechanics apply."
The researchers suggest that "the repetitions in native squid proteins could have a genetic advantage for increased toughness and flexibility."
"We found that the shortest polypeptide chains were brittle," said Demirel. "As they get longer, they are stretchy."
The structural properties in this material are highly programmable. Extremely elastic materials, like the amorphous portion of these proteins, absorb energy and are useful in things like automobile bumpers, while the crystalline portion acts like a spring and is more like the material in a car's dashboard. The proper balance of each could provide the desired materials characteristics.
Their building blocks, the synthetic amino acids, are produced by bacteria so that harvesting of live squid is no longer necessary. Also, the synthetic materials are self-healing, so small cracks and breaks can be repaired. Demirel and his team note that the synthetic mimic of the squid ring teeth proteins can be processed to form a variety of 3-dimensional shapes including ribbons, lithographic patterns and nanotubes with a vast array of potential uses.
More information: Molecular tandem repeat strategy for elucidating mechanical properties of high-strength proteins, PNAS, www.pnas.org/cgi/doi/10.1073/pnas.1521645113
Journal information: Proceedings of the National Academy of Sciences
Provided by Pennsylvania State University