Targeted orphaned domain may lead to drug therapies

By studying an orphaned domain considered disordered by others, Michaela Jansen, Ph.D., associate professor in the Department of Cell Physiology and Molecular Biophysics at the Texas Tech University Health Sciences Center (TTUHSC), may have found the key to possible drug therapies for nervous system diseases and inflammatory processes.
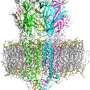
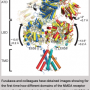
"Pentameric quaternary structure of the intracellular domain of serotonin type 3A receptors," was published in the April issue of Scientific Reports. Along with her research team, Akash Pandhare, Ph.D., and Petar N. Grozdanov, Ph.D., Jansen's research demonstrated for the first time that the serotonin type 3A (5-HT3A) ICD assembles into stable pentamers in solution in the absence of the other two domains, thought to be the drivers for oligomerization.
"We are especially interested in the superfamily of pentameric ligand-gated ion channels (pLGICs) that includes the nAChR, GABA, 5-HT3, and glycine families," Jansen said. "The pLGICs function mainly as neurotransmitter receptors, transforming the chemical signal contained in the neurotransmitter into an electrical signal."
The current therapies make it impossible to precisely hit one target because there are more than 40 different subunits in humans. All the drugs currently in clinical use, target the extracellular or transmembrane domains, which are structurally very similar across different subunits.
Jansen's lab focused on the intercellular domain (ICD), which is diverse. By targeting this domain, the research may lead to new drug therapies without undesired effects. Years ago, the ICD was considered extremely disordered. This research is the first to show the ordered assembly of five ICDs.
"This finding was very unexpected" Jansen said. "Compare this to spaghetti. If you put spaghetti into a boiling pot, you never see groups of five and only groups of five. Many labs research pLGICs, but often remove the ICDs for structural studies because IDCs were believed to be disordered just like spaghettis. Yet this research study found this domain makes very stable arrangements. This is the first indication that it is structurally important, and the first stepping stone for future drug targets."
"Our results suggest the ICD constitutes an oligomerization domain," Jansen said. "This novel role significantly adds to its known contributions in receptor trafficking, targeting and functional fine-tuning. The innate diversity of the ICDs with sizes ranging from 50 to 280 amino acids indicates new methodologies need to be developed to determine the structures of these domains. The use of soluble ICD proteins that we report in the present study constitutes a useful approach to address this gap."
Now with the intracellular domain alone, researchers can develop screening methods to identify small molecules that bind to this domain, as well as study interactions with cytosolic proteins. This domain may have the key to treating diseases like epilepsy, anxiety, Alzheimer's disease and several inflammatory processes.
More information: Akash Pandhare et al, Pentameric quaternary structure of the intracellular domain of serotonin type 3A receptors, Scientific Reports (2016). DOI: 10.1038/srep23921
Journal information: Scientific Reports
Provided by Texas Tech University