May 17, 2016 feature
Dendrite-proof batteries made possible by Zylon nanofibers
(Phys.org)—One of the biggest problems plaguing high-energy, lithium-metal batteries is dendrites, which form when some of the lithium from the electrode begins to branch outside the electrode and into the electrolyte, causing short circuits. In order to control dendrite growth, researchers use membrane separators to try to contain the lithium, but so far no separator material has proved ideal: ceramic separators are brittle and form cracks, polymer separators are mechanically weak, and effective nanofiber separators have so far been very difficult to fabricate.
Now in a new study published in Nano Letters, Xiaoming Hao et al. may have found a separator material that appears to have all the right properties for realizing a dendrite-proof battery. The researchers assembled ultrastrong nanofiber membranes made from a type of polymer microfiber called Zylon (or PBO). Because Zylon is lightweight and even stronger than Kevlar, it has been used in several high-tech applications, including NASA's Martian rovers, space elevator prototypes, and sports equipment such as snowboards and tennis racquets.
Here the researchers showed that, when Zylon microfibers are exfoliated into nanofibers and then cast into thin membranes, the combination of strength, electrical resistance, and high heat tolerance make them exceptionally good membrane separators for preventing dendrite growth in lithium batteries.
"The greatest significance of this work is that ultrastrong nanoporous membranes from exfoliated Zylon nanofibers are developed," coauthor Jian Zhu at Northwestern University in Evanston, Illinois, told Phys.org. "These membranes can serve as high-performance battery separators for suppressing lithium dendrites and tolerating extreme conditions, such as high temperature."
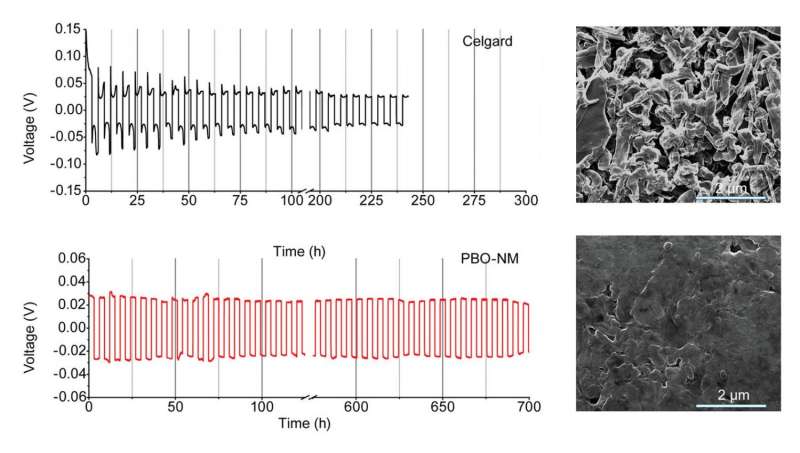
When compared to one of the current state-of-the-art battery separators, Celgard 2400, the new nanofiber membrane exhibits advantages in several areas. After 230 hours of cycling a battery with the Celgard 2400, the electrode surface appears mossy with dendritic lithium growth, whereas an electrode surface with the new nanofiber membrane remains smooth even after 700 hours. This prohibition of dendrite formation results in performance advantages, including higher long-term stability and higher efficiency. The new nanofiber membranes are also more heat-tolerant, so whereas the Celgard 2400 begins to melt at about 125° causing battery failure, batteries with the new membranes continue to operate until 185°, the point at which the electrolyte decomposes.
In the future, the researchers plan to explore strategies for low-cost mass-production of the nanofiber membranes on an industrial scale.
"There are several directions for this area in the future," Zhu said. "One priority is that we are looking for alternative synthesis methods with lower cost and more environmental-friendliness for preparing Zylon (PBO) nanofibers. Our goal is to make these membranes cheap enough to be more attractive for industrial partners. Another direction is the application of these membranes in other battery systems which might involve dendrite formation."
These systems include batteries based on sulfur, sodium, or aluminum, as well as supercapacitors. Due to their exceptional properties, the nanofibers could also be used to reinforce the strength of hydrogels, aerogels, and various composite materials.
More information: Xiaoming Hao et al. "Ultrastrong Polyoxyzole Nanofiber Membranes for Dendrite-Proof and Heat-Resistant Battery Separators." Nano Letters. DOI: 10.1021/acs.nanolett.5b05133
Journal information: Nano Letters
© 2016 Phys.org