Understanding lock for cellular trap door may lead to better disease treatment
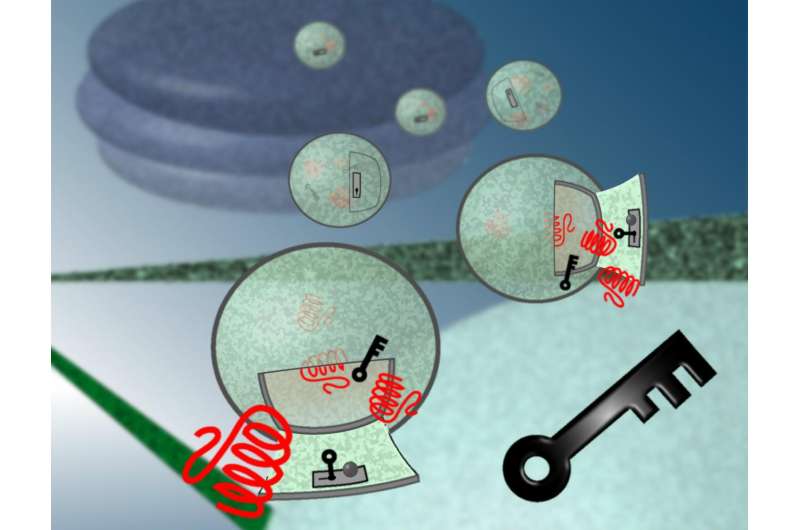
A team of researchers who two years ago announced a "Trojan horse" method of entering a cell without harming it have now found, in effect, the lock to the cellular "trap door."
The research, led by Dr. Jean-Philippe Pellois, appears in the May 5 issue of the journal Cell Chemical Biology.
"We had the key to the cell from the previous research, and now we understand the lock," said Pellois, a Texas A&M AgriLife Research biochemist in College Station.
In 2014, Pellois and his team announced a way to penetrate cellular walls without damaging them—a finding expected to be useful for finding cures and treatments for a wide range of diseases. At the time, Pellois explained that proteins inside cells perform various jobs, so being able to deliver a protein to change or study what was happening inside the cell "would be extremely useful."
Previous methods of crossing a cell's membrane have caused damage or killed the cell, he said. To get proteins inside cells, Pellois and his team used cellular Trojan horses, a trick inspired by the Greek myth in which soldiers concealed themselves inside a statue of a horse so they could sneak into Troy. With this trick, passing through the "cell walls" to deliver the protein directly was more efficient and less harmful, he said.
Still, the researchers needed to understand how the proteins moved from one side of the cell to the other once inside.
"In the Trojan horse example, if you are able to get the horse through the city gate, but what's inside the horse is stuck, it's useless. We knew we had found a key to the Trojan horse trap door, but we didn't know what the key was turning, so to speak, to get into the cell without harming the membrane," Pellois said. "In our most recent work, we looked at where the key was going and what it was doing, and we found the lock."
Because of this work, he said, new sets of keys can now be designed and perfected for various cell biology, biotechnology and therapeutic applications. The interior of a cell also becomes readily accessible and the cell might no longer be an elusive box.
"We can send probes into the cell and spy on the cells to see what the cell is doing inside," he said. "And the lock we found is common across all cell lines. And whereas cell biologists might think that opening the trap door would be toxic to the cell, that has not been so."
He said this finding adds to the potential use by the pharmaceutical industry in designing medicines for better delivery to the source of the disease inside cells.
"In a metabolic disease, an enzyme is mutated or deficient in the cell," Pellois said. "So it would be helpful to be able to replace the defective enzyme with one that is working properly, for example."
He said the technique may eventually be applicable for a host of diseases such as HIV and cancer, and for regenerative medicine for treating heart and liver cells.
Journal information: Cell Chemical Biology
Provided by Texas A&M University