April 28, 2016 report
Nanocrack coating allows membranes to work in high temperature, low humidity environments
(Phys.org)—A team of researchers with members from institutions in South Korea and Australia has developed a coating for membranes used in fuel cells and many other applications that allows it to continue to perform at a high level even as temperatures rise and humidity drops to levels that normally cause performance to suffer. In their paper published in the journal Nature, the team describes their coating, how it works and the different materials that can be improved through its use. Jovan Kamcev and Benny Freeman with the University of Texas at Austin have published a News & Views article in the same journal issue describing the work done by the team and the many ways that the membrane coating has been successfully tested.
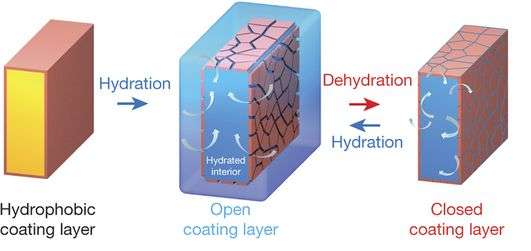
Membranes are a critical part of machines that rely on ionic or size separation—some well-known applications are water filtration efforts, energy generation in fuel cells and flow batteries and by reverse electrodialysis. Though useful, membranes also have a reputation of being rather fragile, resulting in expensive repairs, replacement or performance degradation. One such example is that most membranes need to be kept moist to work properly, which can become problematic in certain environments. Water filtration in a hot Middle Eastern desert, for example, suffers when temperatures soar and humidity levels drop. In this new effort, the research team reports that they have developed a coating for membranes that works similarly to stomatal pores in a cactus plant—the pores open to allow for taking in carbon dioxide during times of higher humidity, such as at night and then close again as the humidity levels drop during the heat of the day.
The membrane coating is made by placing a thin layer of fluorine-related material that is water repellant over the membrane, in a low-humidity environment—under high humidity conditions, nanocracks appear in the material, allowing the water in the air to pass through to the membrane below. But, as temperatures rise and humidity levels drop, the material tightens, closing the gaps where the cracks exist, preventing the water in the membrane from evaporating. Kamcev and Benny Freeman report that the coating has been tested successfully on a wide variety of applications under various environmental conditions, and that thus far, it has proven able to protect delicate membranes in severe environments, allowing for their use in a much broader range to applications.

More information: Chi Hoon Park et al. Nanocrack-regulated self-humidifying membranes, Nature (2016). DOI: 10.1038/nature17634
Abstract
The regulation of water content in polymeric membranes is important in a number of applications, such as reverse electrodialysis and proton-exchange fuel-cell membranes. External thermal and water management systems add both mass and size to systems, and so intrinsic mechanisms of retaining water and maintaining ionic transport1, 2, 3 in such membranes are particularly important for applications where small system size is important. For example, in proton-exchange membrane fuel cells, where water retention in the membrane is crucial for efficient transport of hydrated ions1, 4, 5, 6, 7, by operating the cells at higher temperatures without external humidification, the membrane is self-humidified with water generated by electrochemical reactions5, 8. Here we report an alternative solution that does not rely on external regulation of water supply or high temperatures. Water content in hydrocarbon polymer membranes is regulated through nanometre-scale cracks ('nanocracks') in a hydrophobic surface coating. These cracks work as nanoscale valves to retard water desorption and to maintain ion conductivity in the membrane on dehumidification. Hydrocarbon fuel-cell membranes with surface nanocrack coatings operated at intermediate temperatures show improved electrochemical performance, and coated reverse-electrodialysis membranes show enhanced ionic selectivity with low bulk resistance.
Journal information: Nature
© 2016 Phys.org