Surface chemistry offers new approach to directing crystal formation in pharmaceutical industry
A study by A*STAR researchers suggests the surface properties of the glass vessels in which pharmaceutical ingredients are prepared has an effect on how they crystallize.
When deciding how to control crystallization of an active ingredient during large scale production, drug companies consider many parameters such as solvent type, solute concentration and temperature to ensure the right crystal form.
Different crystal forms (polymorphs) of active pharmaceutical ingredients exhibit different physicochemical properties and can behave very differently once inside the body, so reliable production of the required polymorph is vital.
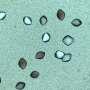
In 2014, Sendhil Poornachary at the A*STAR Institute of Chemical and Engineering Sciences and colleagues showed that the surface chemistry of modified glass vials influences nucleation and growth of selective crystal polymorphs of the anticonvulsant drug carbamazepine. While needle-shaped crystals preferentially formed in the cyano-functionalized vials, tetrahedral-shaped crystals formed in those modified with fluoro- and mercapto-groups. A mixture of the two forms was crystallized inside a control (unmodified) glass vial.
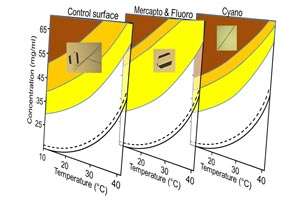
Now, the team has extensively studied the temperature ranges and solute concentrations at which the crystallization of carbamazepine occurs on different chemically-modified surfaces1. By observing the crystal polymorph formed in these vials at a given temperature and concentration, the polymorph occurrence regions were plotted on a temperature–concentration phase diagram (see image). "This type of representation is important to the pharmaceutical industry in the context of defining the design space for a robust crystallization process development," Poornachary says.
The impact of surface chemistry on the crystallization process was then investigated with the help of molecular models. "The experimental results were correlated with the results from a molecular modeling study, which revealed that specific chemical interactions between the crystal structure and functional groups on the template surface promoted nucleation of a particular polymorph," explains Poornachary.
"We envisage that the insights from this work will help develop in silico models to predict the crystallization of [any] active pharmaceutical ingredient on a given template surface," he adds. It is also hoped that surfaces that promote selective nucleation of crystal forms may enable greener, aqueous solvents to be used in a wider range of crystallizations.
Next, the team plans to test the use of functionalized templates to direct the crystallization of carbamazepine on a larger scale. "Potential design approaches for template-induced crystallization process development could include forced flow through functionalized glass capillaries or using functionalized template particles as seeds, rather than the functionalized vessels we have used for this work."
More information: Jose V. Parambil et al. Establishing template-induced polymorphic domains for API crystallisation: the case of carbamazepine, CrystEngComm (2015). DOI: 10.1039/c5ce01080b