Researchers develop nanoporous black gold as light and solid as a bird's bone

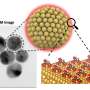
A team of Korean research team, led by Professor Ju-Young Kim (School of Materials Science and Engineering) of Ulsan National Institute of Science and Technology (UNIST), South Korea has announced that they have successfully developed a way to fabricate an ultralight, high-density nanoporous gold (np-Au) material.
In a new paper, published in Nano Letters on March 22, the team reported that this newly developed material, which they have dubbed "black gold" is twice as hard and 30 percent lighter than standard gold.
Prof. Kim says, "This particular nanoporous gold has surface area 100,000 times wider than standard gold. Moreover, due to its chemical stablity, it is also nontoxic to humans."
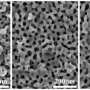
The surface of np-Au is rough and the metal loses its shine and becomes black at sizes less than 100 nanometres (nm).
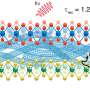
In their study, the team investigated grain boundaries in nanocrystalline np-Au and found a way to overcome the weakening mechanisms of this material, thereby suggesting its usefulness.
The team used a ball milling technique to increase the flexural strength of the three gold-silver precursor alloys. Then, using free corrosion, they dealloyed silver from gold-silver alloys, and were able to produce the nanoporous surface. According to the team, "The size of the pores can be controlled by the temperature and concentration of nitrate." Moreover, they note that these crack-free nanoporous gold samples exhibit excellent durability in three-point bending tests.

Prof. Kim's team says, "Ball-milled np-Au has a much greater density of two-dimensional defects than annealed and prestrained np-Au, where intergranular fracture is preferred. Therefore, the probable existence of a grain boundary opening in the highest tensile region is attributed to the flexural strength of np-Au."
They suggest that this newly developed technique can be applied to many other metals, as the np-Au produced by this technique has increased strength and durability while still maintaining the desirable qualities of standard gold. This means that the technique can also be used in other technologies, substituting for platinum in automobile catalytic conversion, or palladium, in hydrogen sensors.
More information: Eun-Ji Gwak et al. Weakened Flexural Strength of Nanocrystalline Nanoporous Gold by Grain Refinement, Nano Letters (2016). DOI: 10.1021/acs.nanolett.6b00062
Journal information: Nano Letters