Cellular "blindness" to chromosome cohesion defects
Research led by Raquel Oliveira, group leader at Instituto Gulbenkian de Ciência (IGC; Portugal), has elucidated how cells are almost blind to chromosome cohesion defects. The results, published in the scientific journal Cell Reports uncovered how these defects, often associated with cancer development, congenital diseases and infertility, evade the strict surveillance of the checkpoint mechanisms that ensure faithful genome segregation.
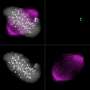
During cell division, the genetic information contained in the chromosomes needs to be faithfully partitioned between the two daughter cells. When the "molecular glue" that holds together the two identical DNA molecules (sister-chromatids) is lost prematurely, chromosomes are distributed randomly leading to daughter cells with a wrong set of chromosomes (also known as aneuploid). It was already known that sister chromatid cohesion defects do not elicit a robust response from the mechanisms that ensure mitotic fidelity. Given the drastic consequences to the cell, this "blindness" appeared paradoxical and the reason for such failure was not fully understood.
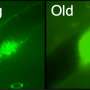
Using fruit flies as a model system (Drosophila melanogaster), combined with a multidisciplinary approach, the researchers triggered artificially premature lost of sister chromatid cohesion and evaluated the behaviour of living cells. Based on experimental observations and mathematical modelling simulations, developed in collaboration with Prof. Bela Novak, University of Oxford, the team concluded that upon premature cohesion loss, cells became gradually less capable of correcting and detecting chromosome attachment errors. "It's like a snow-ball effect: when sister chromatids separate prematurely, the checkpoint mechanisms that govern mitotic fidelity are unable to "read" this mistake in very robust manner. This failure contributes to further weakening of mechanisms that ensure error-correction and checkpoint signalling and consequently the cell is unable to sense that something is wrong", says Raquel Oliveira. This phenomena is explained by Mihailo Mirkovic, first author of the paper: "We uncovered that the mechanisms that detect and correct chromosome attachment defects are ultrasensitive to the activity of CDK1, the master regulator of mitosis. In normal cells this sensitivity ensures faithful segregation of the genetic material. Ironically, this works against the cells if cohesin is lost prematurely, rendering them unable to generate a robust checkpoint response and avoid mitotic errors.
"This work provides an initial theoretical framework to understand how the cell responds to cohesion defects. We are currently investigating, at the organism level, whether the same applies to all cells or if some cells are "blinder" than others, throughout development." says Raquel Oliveira.
More information: Mihailo Mirkovic et al. Premature Sister Chromatid Separation Is Poorly Detected by the Spindle Assembly Checkpoint as a Result of System-Level Feedback, Cell Reports (2015). DOI: 10.1016/j.celrep.2015.09.020
Journal information: Cell Reports
Provided by Instituto Gulbenkian de Ciência (IGC)