Tunable materials clear the way for advanced optics

Now you see it, now you don't. In books and movies, wizards use magic spells to make the visible see-through.
In reality, materials with properties called phase transitions can pull off a similar trick, changing from clear to cloudy depending on the temperature or application of an electric field.
A multi-institutional team of researchers has developed a way to precisely engineer the temperatures at which vanadium dioxide—a material used in high-tech applications ranging from homes to satellites—will undergo phase transition. Their work, published today in the journal Nano Letters, could lead to new types of tunable materials for optics, camouflage and thermal regulation.
"Essentially, any optical component would be better if it were tunable," says Mikhail Kats, senior author of the study and a University of Wisconsin–Madison professor of electrical and computer engineering.
Rather than relying on mechanical components to focus a camera lens or telescope eyepiece, a tunable material could change its innate optical properties on demand.
Scientists have known for more than 50 years that substances like vanadium dioxide can transition between opaque and transparent. However, these materials typically switch under only one particular set of conditions, limiting their applicability.
"In most phase-transition materials, the change occurs at conditions that are far from room temperature, and thus are difficult to incorporate into useful devices," says Kats.
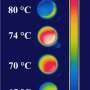
The researchers not only changed vanadium dioxide's intrinsic shift point from 155 degrees Fahrenheit to below 70 degrees, they successfully tuned the transition for that material across a variety of specific temperatures—ranging from typical indoor comfort to medium-rare hamburger.
"This finding is going to open up new frontiers in photonic devices," says collaborator Shriram Ramanathan, a professor of materials engineering at Purdue University.
Additionally, because optical and physical properties arise from the same underlying physical principles, vanadium dioxide's thermal and electrical conductivities also shift with the transition. These types of materials could be used, for example, in homes as "smart" walls or windows that respond to the environment.
"Objects designed to emit light efficiently at high temperatures but not at low temperatures could be used as purely passive temperature regulators that don't require external circuitry or power sources," says Kats.
Materials with this unprecedented versatility could also create new types of thermal camouflage.
"Structures designed to emit the same amount of thermal radiation no matter the temperature could be used to hide objects from infrared cameras," says Kats.
Previously, researchers attempting to change the transition temperatures of vanadium dioxide introduced impurities while trying to uniformly alter the material's entire surface.
Instead, Kats and colleagues bombarded specific regions of the vanadium dioxide with energetic ions. Ion irradiation creates defects in materials, usually an unintended side effect. However, collaborator Carsten Ronning, a professor of solid state physics at the Friedrich Schiller University of Jena in Germany, says the researchers' advance capitalizes on those defects.
"The beauty in our approach is that we take advantage of the 'unwanted' defects," he says.
Directing the ion beam at specific regions of a surface allowed the researchers to make nanoscale modifications to the material.
"We can precisely control the transition temperature everywhere on the sample, with roughly 20-nanometer precision," Kats says. "We have been able to use this method to create effective materials which have multiple phase transitions at the same time."
This technique enabled them to design and create a novel optical polarizer that changes selectivity based on the temperature.
More information: Jura Rensberg et al. Active Optical Metasurfaces Based on Defect-Engineered Phase-Transition Materials, Nano Letters (2016). DOI: 10.1021/acs.nanolett.5b04122
Journal information: Nano Letters
Provided by University of Wisconsin-Madison