Using light for targeted drug delivery could help fight tumors, local infections
Some drug regimens, such as those designed to eliminate tumors, are notorious for nasty side effects. Unwanted symptoms are often the result of medicine going where it's not needed and harming healthy cells. To minimize this risk, researchers have developed nanoparticles that only release a drug when exposed to near-infrared light, which doctors could beam onto a specific site. Their report appears in the Journal of the American Chemical Society.
For years, scientists have been striving to develop localized treatments to reduce side effects of therapeutic drugs. They have designed drug-delivery systems that respond to light, temperature, ultrasound and pH changes. One promising approach involved drug-carrying materials that are sensitive to ultraviolet (UV) light. Shining a beam in this part of the light spectrum causes the materials to release their therapeutic cargo. But UV light has major limitations. It can't penetrate body tissues, and it is carcinogenic with repeated exposure. Near-infrared (NIR) light, however, can go through 1 to 2 centimeters of tissue and would be a safer alternative, but photosensitive drug-carriers don't react to it. Marta Cerruti, Fiorenzo Vetrone and colleagues sought to develop a solution that takes advantage of both types of light.
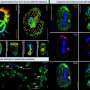
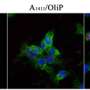
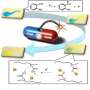
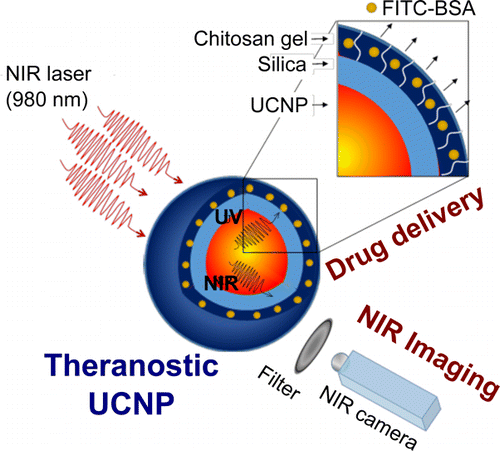
The researchers started with nanoparticles that convert NIR light into UV light and coated them in a UV-sensitive hydrogel shell infused with a fluorescent protein, a stand-in for drug molecules. When exposed to NIR light, the nanoparticles instantaneously converted it to UV, which induced the shell to release the protein payload. The researchers note that their on-demand delivery system could not only supply drug molecules but also agents for imaging and diagnostics.
More information: Ghulam Jalani et al. Photocleavable Hydrogel-Coated Upconverting Nanoparticles: A Multifunctional Theranostic Platform for NIR Imaging and On-Demand Macromolecular Delivery, Journal of the American Chemical Society (2016). DOI: 10.1021/jacs.5b12357
Abstract
Lanthanide-doped upconverting nanoparticles (UCNPs) have emerged as excellent nanotransducers for converting longer wavelength near-infrared (NIR) light to shorter wavelengths spanning the ultraviolet (UV) to the visible (Vis) regions of the spectrum via a multiphoton absorption process, known as upconversion. Here, we report the development of NIR to UV–Vis–NIR UCNPs consisting of LiYF4:Yb3+/Tm3+@SiO2 individually coated with a 10 ± 2 nm layer of chitosan (CH) hydrogel cross-linked with a photocleavable cross-linker (PhL). We encapsulated fluorescent-bovine serum albumin (FITC-BSA) inside the gel. Under 980 nm excitation, the upconverted UV emission cleaves the PhL cross-links and instantaneously liberates the FITC-BSA under 2 cm thick tissue. The release is immediately arrested if the excitation source is switched off. The upconverted NIR light allows for the tracking of particles under the tissue. Nucleus pulposus (NP) cells cultured with UCNPs are viable both in the presence and in the absence of laser irradiation. Controlled drug delivery of large biomolecules and deep tissue imaging make this system an excellent theranostic platform for tissue engineering, biomapping, and cellular imaging applications.
Journal information: Journal of the American Chemical Society
Provided by American Chemical Society