Solid electrolyte interphases on lithium metal anode
The prestigious Advanced Science journal has just published a review paper on solid electrolyte interphases of lithium metal anodes contributed by Prof. Qiang Zhang in Tsinghua University, China and Ji-Guang Zhang in Pacific Northwest National Laboratory.
"Lithium (Li) metal is regarded as the 'Holy Grail' of rechargeable battery technologies due to the high theoretical specific capacity, 3860 mA h g-1, 10 times that of commercial graphite anode, and the lowest redox potential, -3.040 V vs. the standard hydrogen electrode. Therefore, lithium metal batteries, such as lithium-sulfur and lithium-oxygen batteries with the theoretical energy density of 2600 and 3400 Wh kg-1, could be promising candidates in next-generation energy storage devices," said Dr. Qiang Zhang, an associate professor at Department of Chemical Engineering, Tsinghua University, Beijing, China.
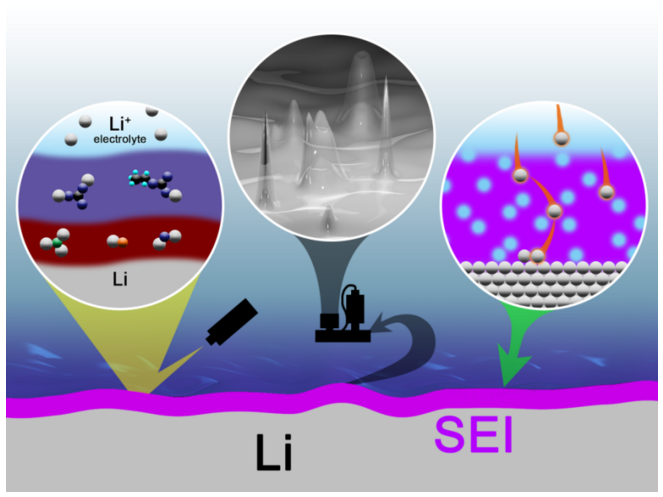
"However, the safe use of lithium metal as an anode is still a great challenge, for the dendritic and mossy metal deposits are very easily obtained on working lithium metal anode. Lithium dendrites induce a low Coulombic efficiency and severe safety risk, hindering the practical demonstration of high-energy-density lithium metal batteries. The dendrite nucleation and growth are closely related to the surface layer between the electrolyte and anode, called the solid electrolyte interphase (SEI). The surface component and structure of the SEI layer play an extremely important effect on the morphology of lithium deposits and decide the cycling performance of lithium metal anode. Consequently, it is of great importance to have a deep understanding on the SEI layer."
"The recent achievements on the SEI layer of the lithium metal anode are highlighted in my manuscript," said Xin-Bing Cheng, a graduate student and the first author of the review, "We have briefly summarized the mechanisms of SEI formation and models of SEI structure. The analysis methods to probe the surface chemistry, surface morphology, electrochemical property, dynamic characteristics of the SEI layer are emphasized. The critical factors affecting the SEI formation, such as electrolyte component, temperature, current density, are comprehensively debated. The paper summarizes efficient methods to modify the SEI layer with the introduction of a new electrolyte system and additives, ex-situ formed protective layer, and electrode design."
Although these works afford new insights into SEI research, a robust and precise route for SEI modification with a well-designed structure, as well as the relationship between structure, properties, and electrochemical performance, is still inadequate. More studies on SEI layer building require collaborative works from physics, chemistry, nanomaterials, and engineering communities.
"Through further investigation on the science and engineering of SEI on lithium metal, the use of lithium metal as a superior anode in a rechargeable cell is quite promising. The ultra-stable and robust SEI will enable broad applications of rechargeable Li metal in advanced Li–S batteries, Li–air batteries, and other advanced Li batteries," Qiang told Phys.org.
More information: Xin-Bing Cheng et al. A Review of Solid Electrolyte Interphases on Lithium Metal Anode, Advanced Science (2015). DOI: 10.1002/advs.201500213
Provided by Tsinghua University