Toward tires that repair themselves
A cut or torn tire usually means one thing—you have to buy a new one. But some day, that could change. For the first time, scientists have made tire-grade rubber without the processing step—vulcanization—that has been essential to inflatable tires since their invention. The resulting material heals itself and could potentially withstand the long-term pressures of driving. Their report appears in the journal ACS Applied Materials & Interfaces.
Vulcanization involves adding sulfur or other curatives to make rubber more durable while maintaining its elasticity. But once an errant piece of glass or other sharp object pierces a tire, it can't be patched for long-term use. Researchers are beginning to develop self-healing rubber in the laboratory, but these prototypes might not be stable over time either. Amit Das and colleagues wanted to address that shortcoming.
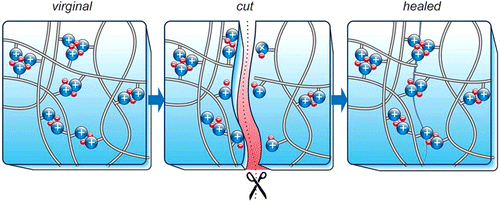
Using a new simple process that avoids vulcanization altogether, the researchers chemically modified commercial rubber into a durable, elastic material that can fix itself over time. Testing showed that a cut in the material healed at room temperature, a property that could allow a tire to mend itself while parked. And after 8 days, the rubber could withstand a stress of 754 pounds per square inch. Heating it to 212 degrees Fahrenheit for the first 10 minutes accelerated the repair process. The researchers say their product could be further strengthened by adding reinforcing agents such as silica or carbon black.
More information: Ionic Modification Turns Commercial Rubber into a Self-Healing Material, ACS Appl. Mater. Interfaces, 2015, 7 (37), pp 20623–20630 DOI: 10.1021/acsami.5b05041
Abstract
Invented by Charles Goodyear, chemical cross-linking of rubbers by sulfur vulcanization is the only method by which modern automobile tires are manufactured. The formation of these cross-linked network structures leads to highly elastic properties, which substantially reduces the viscous properties of these materials. Here, we describe a simple approach to converting commercially available and widely used bromobutyl rubber (BIIR) into a highly elastic material with extraordinary self-healing properties without using conventional cross-linking or vulcanising agents. Transformation of the bromine functionalities of BIIR into ionic imidazolium bromide groups results in the formation of reversible ionic associates that exhibit physical cross-linking ability. The reversibility of the ionic association facilitates the healing processes by temperature- or stress-induced rearrangements, thereby enabling a fully cut sample to retain its original properties after application of the self-healing process. Other mechanical properties, such as the elastic modulus, tensile strength, ductility, and hysteresis loss, were found to be superior to those of conventionally sulfur-cured BIIR. This simple and easy approach to preparing a commercial rubber with self-healing properties offers unique development opportunities in the field of highly engineered materials, such as tires, for which safety, performance, and longer fatigue life are crucial factors.
Journal information: ACS Applied Materials and Interfaces
Provided by American Chemical Society


















