Preventing chromosomal chaos: Protein-based genome-stabilizing mechanism discovered
Most people are familiar with the double-helix shape that allows genetic information to be packed into a molecule of human DNA. Less well-known is how all this information - which, if laid end-to-end, would stretch some three meters - is packed into the cellular nucleus. The secret of how this crush of genetic code avoids chaos - remaining untangled, correctly compartmentalized, and available for accurate DNA replication - has recently been revealed.
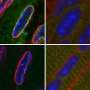
By tracking and analyzing the movement of fluorescently-tagged genomic regions within the nuclei of live cells, an international team has determined that a protein known as lamin A plays a central role in maintaining genomic structural stability.
The researchers - led by Prof. Yuval Garini of Israel's Bar-Ilan University, and with the participation of members of his lab including Dr. Irena Bronshtein-Berger and Dr. Eldad Kepten, then a PhD candidate - have shown how this protein is involved in the formation of "cross-links" that limit genetic material's freedom of movement within the nucleus. This creates a stable and linked polymeric structure that promotes chromosomal integrity and makes normal cellular replication possible.
The team's findings - published on August 24th, 2015 in Nature Communications - present a comprehensive model for explaining the biophysical underpinnings of chromosome dynamics and organization. The study also provides intriguing insight into the pathology of a number devastating human diseases associated with a mutated form of the lamin A protein.
An "Anchor" for Chromatin Organization
"In our lab, we study chromatin - the complex of DNA and proteins that form the chromosome structure within the nucleus," says Garini, a member of Bar-Ilan University's Department of Physics who also heads the University's Institute of Nanotechnology and Advanced Materials. "Chromatin moves dynamically inside the nucleus while, at the same time, remains highly ordered. Our study shows, for the first time, how chromatin movement is regulated by proteins, and how this regulation affects function."
According to Garini, this protein-mediated mechanism is fundamental to the stability of life as we know it.
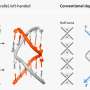
"Inside the nucleus, extremely long DNA strings create what might be described as a bowl of dynamically-jiggling 'genetic spaghetti'," Garini says. "If this material becomes tangled or knotted, ordered DNA duplication - an important step in normal cell division - cannot occur. Using advanced microscopic techniques, we determined that genomic organization is controlled by inter-chromosomal binding events that 'anchor' chromatin regions to each other to achieve flexible stability."
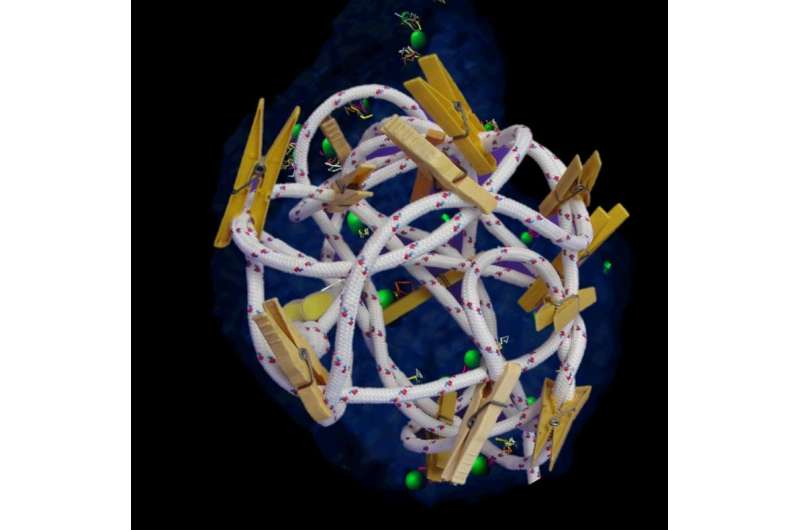
To help non-scientists visualize what "flexible stability" looks like, Garini suggests the following. "Imagine a length of rope that is randomly compressed into a small space. If, at various points along the rope's length, the rope is linked with clips to other points on the rope, this creates a slightly flexible, yet stably-ordered structure. This stability - created by inter-chromosomal binding events - is what allows chromatin order to be maintained."
Garini says it is not yet clear whether lamin A is solely responsible for stabilizing chromatin structure, or if credit should go to some larger network of associated proteins with which lamin A is associated. However, he asserts, the study does reveal the heretofore unrecognized role of lamin A in chromatin dynamics.
Earlier studies of chromatin have suggested that structural organization may be mediated by the interaction of specific chromosomal domains with the nuclear envelope. However, in the current study, Garini reports that a significant fraction of lamin A's activity occurs throughout the nucleoplasm, demonstrating that this nucleus-wide activity bears significant responsibility for chromatin's structural stability.
Support for the new model was provided by Garini's observation of dramatically different chromatin dynamics when lamin A was removed.
"We engineered lamin-A-deficient cells, and then measured the speed and range of chromatin movement," he says. "As opposed to the slow and localized movement characteristic of cells that contain the lamin A protein, in the deficient cells we observed motion that was fast and highly-dispersed. Such fast and loose chromatin dynamics are a perfect recipe for genetic confusion - and hereditary chaos."
Single-Particle Tracking of Nuclear "Traffic Patterns"
The researchers employed a variety of techniques - drawn from the worlds of biophysics, mathematics, advanced microscopy and genetic engineering - to quantify chromatin dynamics in the presence and absence of the lamin A protein.
"Single particle tracking is a technique that makes it possible to label almost any point in a live cell with a fluorescent marker, then create a 'movie' that reveals its changing position by capturing a series of images over time. In the current study, rather than focusing on just one moving point, we analyzed the simultaneous movement of many DNA sub-units to characterize overall patterns," Garini explains, adding that this methodology is analogous to the traffic analyses created by WAZE and other network-based navigation devices.
"By following hundreds of paths - and by analyzing the results with mathematical tools - some of which were created in our lab - we were able to clarify how the presence of lamin A constrains chromatin movement and promotes genomic organizational stability."
Medical Ramifications
The stability-promoting mechanism discovered by Garini and his colleagues - unlike any model of chromatin organization ever put forward - may have significant implications for the understanding and clinical treatment of disease. Specifically, it may pave the way to new approaches toward conditions associated with mutations in the lamin A protein.
"One disease linked to mutated lamin A is progeria - a rare but heartbreaking disorder that causes children to age rapidly, beginning in their first years of life," Garini says. "Although we have not studied this disease in our lab, it may be possible that the lamin A mutation which is a hallmark of the disease prevents cells from dividing in an orderly way, as explained above."
Mutated lamin A is also a hallmark muscular dystrophy. "It may be that genetic screening for this mutation could someday be used for medical diagnostics," Garini says. "It may also be possible that the new model revealed by our study may help eventually support targeted drug development."
An Unusual, Biophysical Approach
In the meantime, Garini will continue to use the tools of biophysics to characterize how chromatin organization and dynamics contribute to genome-wide stability - an approach that he says is relatively unusual.
"Despite an enormous amount of important research in genetics, very few labs have focused on characterizing how our genetic material is organized in the nucleus, and how this organization contributes to function," he says. "Now, we have a very reasonable explanation - supported by a significant number of observations for how genomic structure is conserved. If this stabilizing mechanism did not exist, our genes - the 'code of the program' that defines life as we know it - might never be passed down to the next generation."
Journal information: Nature Communications
Provided by Bar-Ilan University