Mathematical kinetics model accurately depicts grain-level corrosion plaguing metal alloys
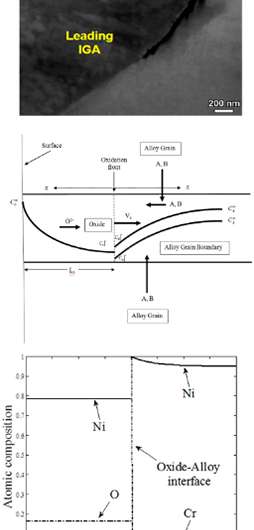
A core concern for any structural material is maintaining a prolonged, effective service life. Notably, critical metal alloy failures typically initiate beyond the naked eye, where intergranular (between the grains) oxidation combines with stress corrosion, causing cracks. Even worse, these corrosive attacks ultimately can result in total metal alloy failure. In their work examining intergranular attack of alloys under hydrothermal conditions, scientists from PNNL's ACMD Division Computational Mathematics group, Physical Sciences Division, and Energy and Environment Directorate developed a mathematical model that is directly comparable to experimental data in predicting how fast oxygen penetrates binary alloys and the resulting depletion of select elements in the materials that leads to failures. Their work reveals insights into oxidation mechanisms at the atomic level that provide a new perspective on ways to improve materials durability.
Combating intergranular embrittlement and disintegration is necessary for preventing metal alloy failure during service life, yet oxidation remains a potentially dangerous obstacle to overcome. Understanding the kinetics and mechanisms causing intergranular oxidation at the atomic level can improve manufacturing for more durable, corrosion-resistant materials designed for safe application in service environments, for example, in a light-water (nuclear) reactor within its decidedly high-temperature, reactive environs.
In seeking a mathematical kinetics model to predict oxidation penetration and minor element depletion, the PNNL team began with a generic model and earlier studies that involved moving interfaces, oxidation kinetics, and heterogeneous reactions at the interface of two phases. To fully examine intergranular oxidation rates, they noted that the transport processes along the grain boundary and reactions at the oxidation front had to be considered. Using a semi-analytical method to find numerical solutions of the proposed model, they approximated various real-world scenarios. For planar (flat) surface oxidation, the model predicts a critical concentration, where the oxide of the minor element dominates the oxide of the major element by neglecting mass transfer between the bulk alloy and grain boundary. Meanwhile, for grain boundary oxidation, the model can account for the penetration velocity, penetration depth, and depletion distance ahead of the advancing oxidation front based on the competition of oxidation and transport properties of various species.
The proposed selective oxidation model provides a simple theoretical framework for understanding the complex picture of oxidation and transport during intergranular attacks. Future work includes building a multiscale simulation framework that establishes connections between the proposed model to atomistic models based on explicit parametrization of relevant transport and oxidation parameters via appropriate small-scale models.
More information: "Modeling selective intergranular oxidation of binary alloys." The Journal of Chemical Physics 142(1):014704. DOI: 10.1063/1.4905226
Journal information: Journal of Chemical Physics
Provided by Pacific Northwest National Laboratory



















